The Muscle Problem With Ozempic Has a Fix.
Bimagrumab preserves the muscle that semaglutide takes with the fat.
Weight loss drugs are judged by the number on a scale. But the scale is a crude instrument. It makes no distinction between fat and muscle. Up to a third of the weight lost on Ozempic is muscle. That ratio holds across the major GLP-1 trials. Semaglutide and Tirzepatide drive approximately 15 to 22 percent total body weight loss (Wilding et al., 2021; Jastreboff et al., 2022), but lean tissue accounts for a quarter to a third of it (Heymsfield et al., 2014). Strength is a predictor of longevity (García-Hermoso et al., 2018). For older adults with sarcopenia or anyone already carrying low muscle mass, the additional loss is its own risk. The field has known for years, and it was quietly downplayed. Because there was no fix.
A phase 2 trial published in Nature Medicine provides a solution (Heymsfield et al., 2026). The drug is bimagrumab, a monoclonal antibody that blocks activin type II receptors. Combined with semaglutide, it produced more total weight loss while preserving the lean mass that semaglutide strips away.
If you enjoy these posts, consider subscribing and becoming a part of our growing community!
Bimagrumab was built to treat muscle wasting, not obesity (Heymsfield et al., JAMA, 2021). Its target, the activin type II receptor, sits on the surface of muscle and fat cells. It receives signals from myostatin, activin A, activin E, and several other TGF-beta family ligands that suppress muscle growth and regulate fat storage. Bimagrumab blocks the receptor itself, shutting off the entire suppressive input. That is why it works where earlier approaches failed. Myostatin knockout mice are enormous. Belgian Blue cattle carry a natural myostatin loss-of-function. But every drug built to block myostatin alone has failed in humans. Stamulumab, domagrozumab, landogrozumab. Two decades of clinical attempts, none of them worked. Myostatin is one of several signals converging on the same receptor. Blocking one ligand was never enough. Bimagrumab blocks the receptor itself and catches them all.
The effect on fat was unexpected. Exome sequencing from the UK Biobank identified variants in the activin E and ALK7 genes that protect against abdominal obesity. Bimagrumab blocks the same signaling node, increasing fat mobilization independently of appetite.
The BELIEVE trial confirmed this directly. Caloric intake in the semaglutide groups dropped. In the bimagrumab groups, it did not. Bimagrumab reshapes body composition without changing how much patients eat. Two drugs, two tissues, two completely different pathways. One works through the brain. The other works through muscle and fat.
The BELIEVE trial randomized over 500 adults with obesity across nine arms, including bimagrumab alone, semaglutide alone, and four dose combinations. At 48 weeks, the high-dose combination (bimagrumab 30 mg/kg plus semaglutide 2.4 mg) produced 17.8 kg of total weight loss versus 14.2 kg for semaglutide alone.
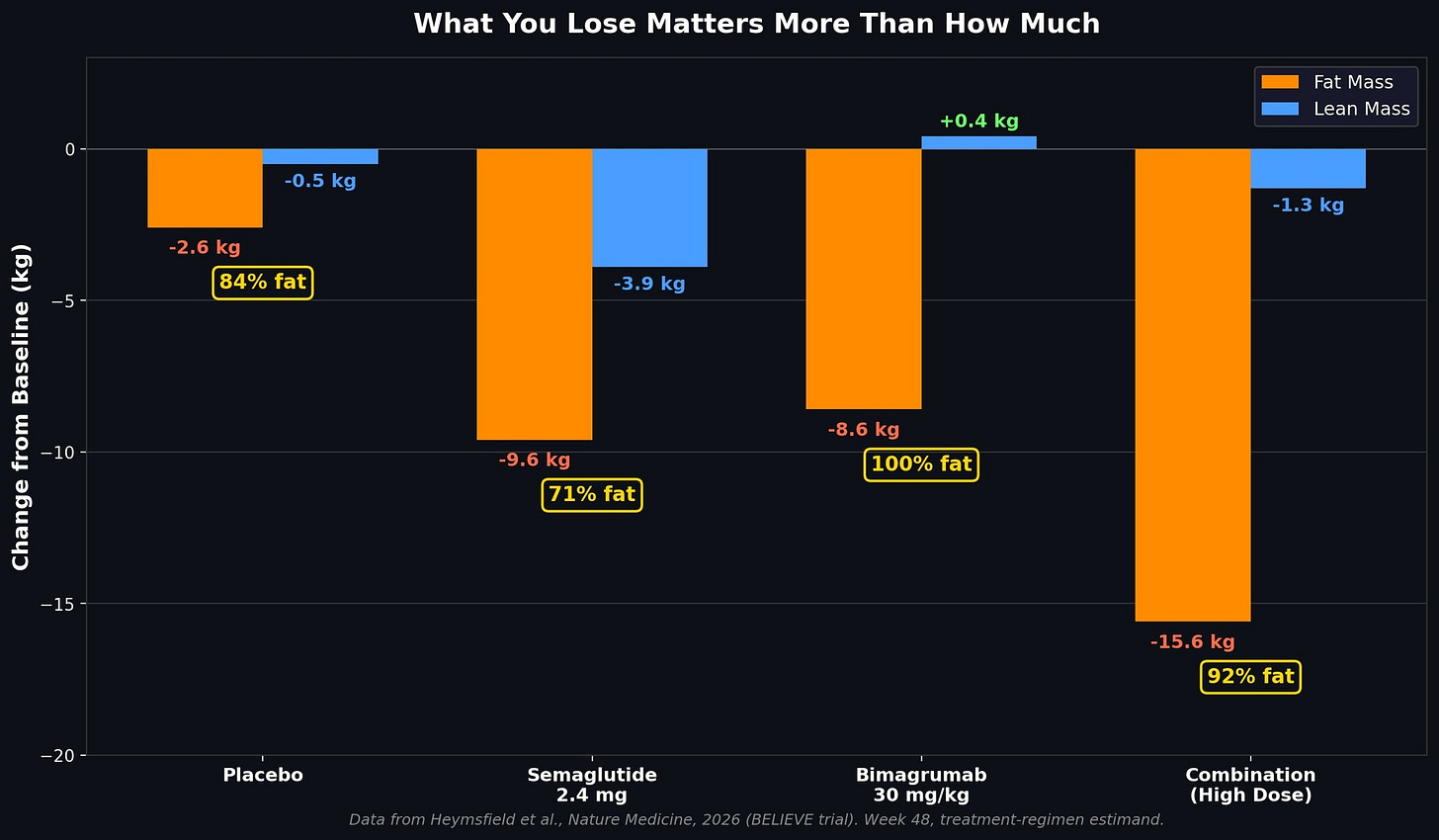
On semaglutide alone, roughly a third of the weight lost was muscle. In the high-dose combination, almost none of it was. Ninety-two percent of the combination’s weight loss was fat. Bimagrumab alone went further. It lost fat and gained lean mass. Every kilogram it shed was fat. Losing fat while gaining muscle is the one thing diet and exercise cannot reliably do at the same time. Bimagrumab did it by default.
By week 72, the gap widened. Semaglutide alone had shed 7.4 percent of lean tissue. Bimagrumab alone had gained 4.1 percent. The combination held lean mass near baseline while losing nearly half its visceral fat.
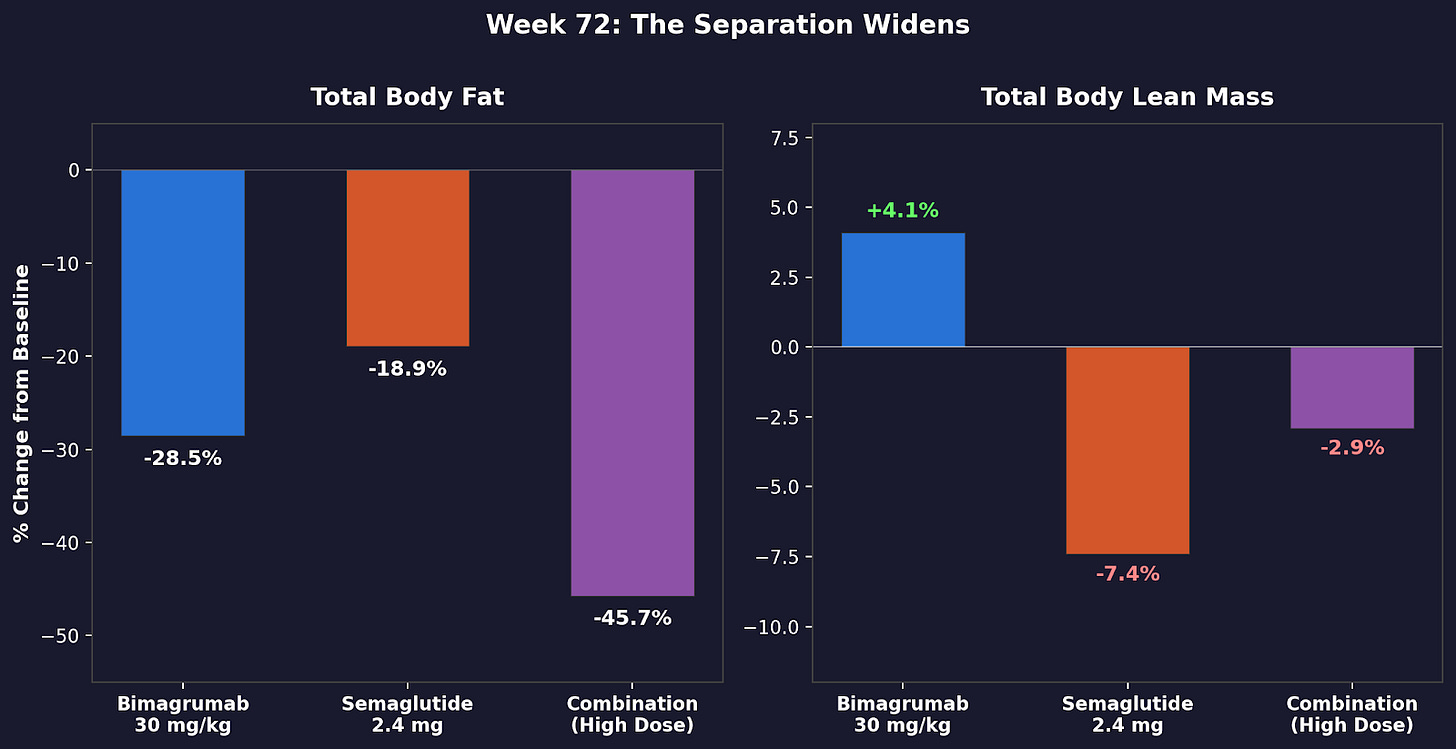
Grip strength increased in bimagrumab-containing groups, confirming the lean mass numbers translate to function. Grip strength is also an independent predictor of longevity, particularly in women (BioWire Byte 021).
Every prediabetic patient in the high-dose combination normalized blood sugar by week 72. One hundred percent.
The safety signals were predictable. Muscle spasms occurred in roughly two-thirds of bimagrumab patients. Acne was common. LDL cholesterol rose with bimagrumab while semaglutide lowered it. The combination partially offset the rise but did not eliminate it. Discontinuation rates were lower in the combination arms than in bimagrumab alone, suggesting semaglutide may improve tolerability. (The trial was funded by Eli Lilly, which acquired bimagrumab’s developer Versanis Bio.)
This is phase 2. It was built to find doses and characterize the biology, not to prove efficacy at scale. Phase 3 will need larger populations, longer treatment, and MRI confirmation of the DXA-based body composition data, particularly in older adults with the most to lose. A trial combining bimagrumab with tirzepatide is already registered.
GLP-1s solved appetite. Activin receptor blockade may solve body composition. If this combination delivers in phase 3 what it delivered here, it will redefine what counts as successful weight loss. Not just pounds lost. Pounds of what?
These newsletters take significant effort to put together and are totally for the reader’s benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References
Heymsfield SB, Aronne LJ, Montgomery P, et al. Bimagrumab plus semaglutide alone or in combination for the treatment of obesity: a randomized phase 2 trial. Nature Medicine. 2026;32:869-882. doi: 10.1038/s41591-026-04204-0
Heymsfield SB, Gonzalez MC, Shen W, Redman L, Thomas D. Weight loss composition is one-fourth fat-free mass: a critical review and critique of this widely cited rule. Obes Rev. 2014;15:310-321. doi: 10.1111/obr.12143
Heymsfield SB, Coleman LA, Miller R, et al. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA Netw Open. 2021;4(1):e2033457. doi: 10.1001/jamanetworkopen.2020.33457
Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989-1002. doi: 10.1056/NEJMoa2032183
Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-216. doi: 10.1056/NEJMoa2206038
García-Hermoso A, Cavero-Redondo I, Ramírez-Vélez R, et al. Muscular strength as a predictor of all-cause mortality in an apparently healthy population: a systematic review and meta-analysis of data from approximately 2 million men and women. Arch Phys Med Rehabil. 2018;99(10):2100-2113. doi: 10.1016/j.apmr.2018.01.008




Not in pharmacology ever since working in Life Zciences. I am interested in if there are tools for modeling efficacy.
The weight loss drugs that suppress appetite don't address one of the major contributors to weight gain, which is eating from stress or boredom. I think that for any weight loss drug to be really effective, regardless of its mechanism of action, it would have to be accompanied by at least a little cognitive therapy so people substitute mindfulness for mindless grazing. You're bored before bedtime and it's easy to start snacking. Recognizing why you reach for the chips can help you redirect and form a new, more constructive response. If you're going to get off the couch and walk into the kitchen, why not walk the other way, instead, and do something productive? Everyone defines productivity differently, but as long as it doesn't include eating more you'll probably make more weight loss progress than with drugs alone or in combination.