VIII. Biological Strategies to Expand Human Intelligence: Neurogenesis
Mitigating the Existential Risk of AI: Interventions to Enhance Human Intelligence
I. Overview and Introduction | II. The Inevitability and Existential Risk of Artificial General Intelligence | III. Understanding Human Intelligence | IV. Reaching vs. Expanding Biological Potential | Biological Strategies to Expand Human Intelligence: Neurotransmitter Modulation | VI. Biological Strategies to Expand Human Intelligence: Neurotrophins | VII. From Neurons to AI: The Surprising Symmetry of Emergence | VIII. Biological Strategies to Expand Human Intelligence: Neurogenesis
FOREWORD
In this series of essays, we discuss the existential risk posed by general AI eclipsing human intelligence. It is for these reasons that it is necessary for us to similarly scale biological intelligence to maintain mankind's autonomy, purpose, and possibly survival into the future. We have begun discussing cognitive enhancement strategies and their potential implementation to improve biological intelligence. Thus far, these strategies focused on manipulating the signaling between neurons or the molecules governing neuron growth and health. This post will continue that discussion with the introduction of neurogenesis, the birth of new neurons, and assess how this strategy may potentially be the holy grail for intelligence advancements.
NEUROGENESIS INTRODUCTION AND OVERVIEW
In a previous essay, we observed and discussed how intelligence across species correlates positively with cortical neuron number. This is to say, more neurons, the fundamental units of the brain’s circuitry, typically translate to more intelligent behavior, e.g., the ability to reason and solve problems. This seems fairly intuitive. One would expect that as the number of subunits for information processing and storage capability increases, so should the ability to store and process information, i.e., more neurons being equivalent to more transistors on a computer chip. Furthermore, as we discussed, new properties can emerge as systems become larger and more complex. But does this same principle hold true within our own species as well?
The next inference one might make is that human intelligence may similarly increase with additional neurons. While neuron count is not the full story since their configuration and interconnectedness are impactful, the cortice size still does play an important role. So, the critical question becomes: how do we increase cortical neuron density? Where would these new neurons originate? Unlike most cells , which replicate through mitosis (cell division), neurons follow a unique path of renewal.
Neurons stand out in their inability to divide. Our brain’s functionality is a result of an elaborate network formed by axons (for sending signals), dendrites (for receiving signals), and synapses (where signals are relayed). These neural connections are woven and reinforced over years of environmental conditioning. The process of cell division would disrupt this architecture and the information stored within. Yet, neurons are not immortal; they are gradually replaced throughout our lives, a process known as neurogenesis.
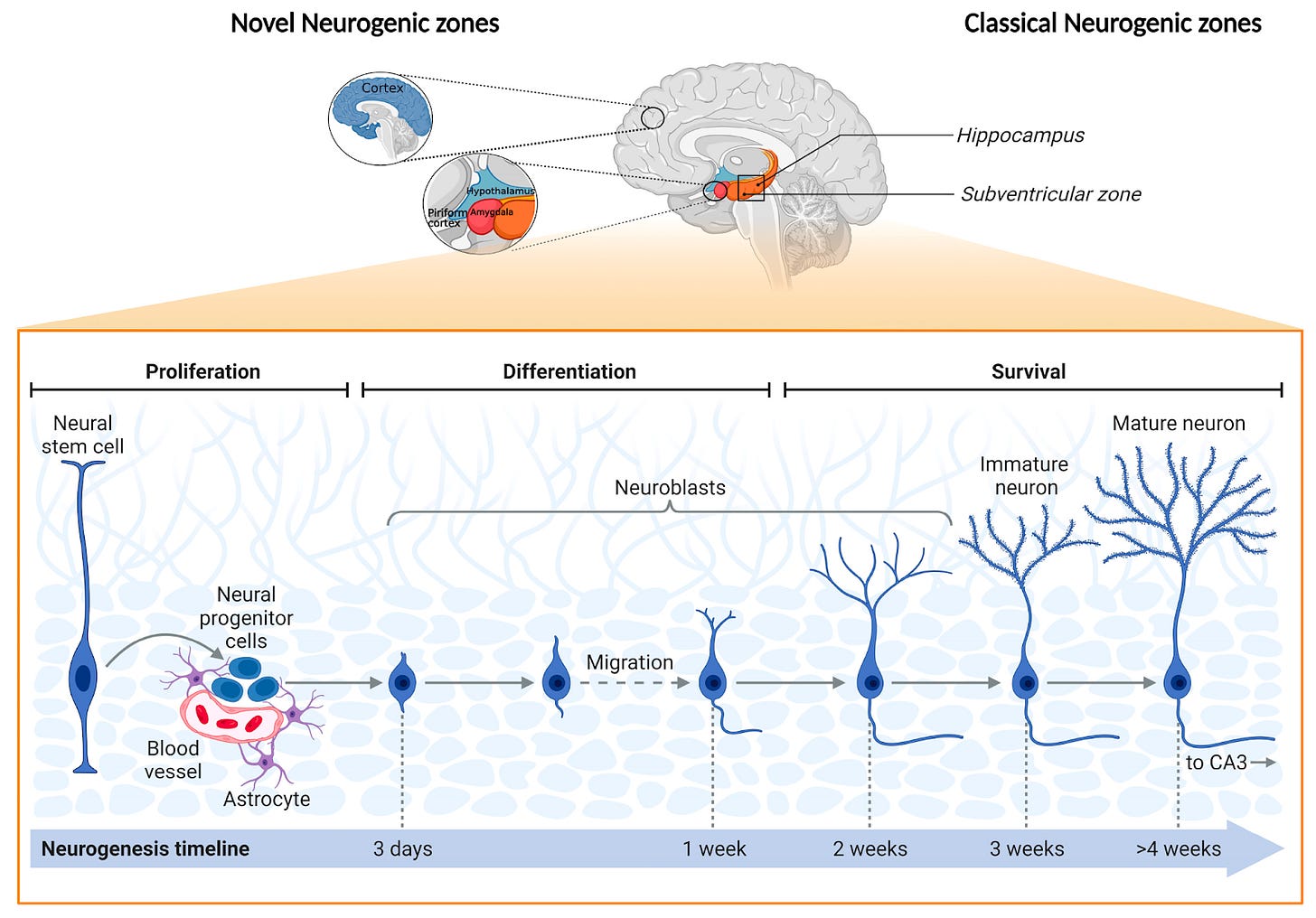
Neurogenesis is the birth of new neurons from neural stem cells or progenitor cells (Figure 1). This offers an exciting cognitive enhancement opportunity for many areas such as memory and learning. The number of neurons in the human brain remains relatively stable from birth to adulthood. It is the connections between these neurons that develop as we mature, learn, and gain new experiences. While mitosis could disrupt the complex networks neurons form, neurogenesis persists in areas like the hippocampus, regulated by growth factors, neurotransmitters, and environmental factors. Tapping into this process for producing new neurons and potentially meaningful cognitive enhancement, however, remains a formidable scientific challenge, paving the way for our next exploration.
Mechanism of Neurogenesis: a closer look
In our attempt to understand how we might increase cortical neuron density, we arrive at the fascinating biological process known as neurogenesis. But where does neurogenesis take place and how does it actually work? What are the cellular and molecular events that lead to the birth of new neurons? Let's delve into the details.
The starting line: neural stem cells are the starting material for new neurons. The story begins with neural stem cells (NCSs) that are believed to be tucked away in specialized regions of the brain like the subventricular zone and the hippocampus (Figure 1). These cells are multipotent, meaning they have the potential to give rise to various types of neural cells, including neurons, astrocytes, and oligodendrocytes. Within these NSC reservoirs, specialized signaling molecules are secreted to maintain the NSCs, replenish their population, or recruit them into new neurons (Silva-Vargas, 2016). In essence, they're the "parent" cells waiting for the right signals to differentiate into "offspring" neurons. Studies as early as 1995 have shown that neural stem cells are not just a feature of the developing brain but are present throughout life (Gage et al, 1995).
The green light: neurogenesis begins with signaling to neural stem cells. For neural stem cells to kickstart the process of becoming a neuron, they need a green light. This “go” signal comes either by an extrinsic environmental influence or intrinsic genetic factors. In the case of extrinsic stimulation, this occurs by secreted signaling molecules in the local microenvironment, e.g., morphogens, growth factors, and neurotransmitters (Faigle et al, 2013). When these molecules bind to specific receptors on the neural stem cell, they trigger a cascade of intracellular events that push the cell toward becoming a neuron. The secreted factors are believed to play a predictive role in the ultimate differentiation of the NSC to specific specialized neural subtypes (Mu et al, 2010). Alternatively, intrinsic genetic mechanisms can arise internally within the cell by transcription factors or epigenetic regulators. Much of these processes are still being understood.
The roadmap: neural stem cell differentiation to terminal neurons is guided by gene expression. Once the green light is given, the cell's machinery starts to express specific genes that will guide its transformation into a neuron. This involves synthesizing proteins that are essential for neural function, such as ion channels for electrical signaling and synaptic proteins for communication with other neurons. Studies have mapped out the gene expression profiles that are crucial for neural differentiation (Okano et al, 2015).
The final form: New neurons must migrate to a new home and integrate into existing neural circuitry. After differentiation, the young neurons, often called "neuroblasts," still have a long journey ahead. They need to mature, extending axons and dendrites to make connections with existing neurons. This is a highly regulated process, as the new neurons must integrate seamlessly into existing neural circuits without causing disruptions. They form synapses, start firing action potentials, and essentially become part of the brain's communication network. Research has shown that newly formed neurons can integrate into existing neural networks and contribute to brain function (Toni et al, 2007).
The Checks and Balances: Inhibitory Signals. It's worth noting that not all neural stem cells receive the go-ahead to become neurons. Some are inhibited by signaling molecules that maintain them in a stem cell state. This balance between differentiation and maintenance is crucial for the brain's stability and is an area of active research (Encinas et al, 2011).
Understanding these mechanisms is not just an academic exercise; it has real-world applications. For instance, manipulating these pathways could lead to treatments for neurodegenerative diseases or even methods for cognitive enhancement. My interest in this research primarily focuses on the latter.
IMPACTS ON INTELLIGENCE
Now that we understand the basic mechanism of how the body can regenerate neurons through neurogenesis, how can this translate to changes in intelligence? Moreover, what specific traits associated with general intelligence are affected by the integration of new neurons? We’ll discuss a few of these below.
Memory Enhancement: The hippocampus, a region where neurogenesis is known to occur in adults, plays a crucial role in memory formation. While the mechanisms of memory are not perfectly understood, it is believed that configurations of neurons encode, store, and recall memories. However, as we age, we begin to reach capacity in the ways we can store new information, which can require removing older information. New neurons have the potential to enhance the volume of memory retention and recall through the creation of additional neural configuration patterns (Figure 2).
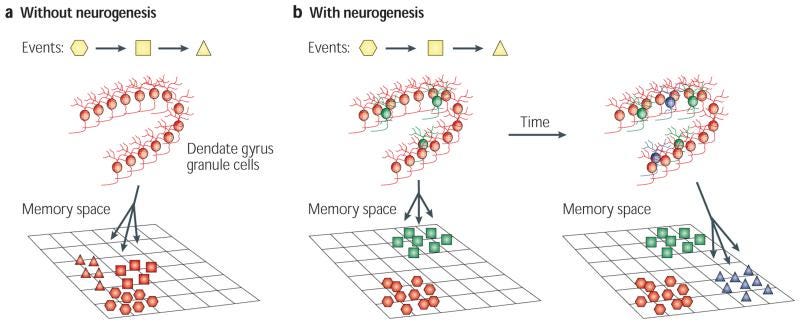
Pattern separation and Cognitive Flexibility: New neurons are believed to be more plastic, meaning they can form connections more easily than older neurons. This could lead to increased cognitive flexibility, allowing individuals to adapt and learn new information more efficiently. For example, in rodent studies, experimentally reducing endogenous neurogenesis results in a poorer ability to discriminate between highly similar patterns, whereas increasing the number of new neurons produced results in an increased ability to successfully discriminate between highly similar stimuli (Clelland et al, 2009; Sahay et al, 2011). This is fitting with the hypothesis that biological neuronal networks are tiny pattern recognition units and additional networks would enable more and faster recognition.
OPPORTUNITIES
Endogenous neurogenesis:
Endogenous neurogenesis is the body's natural ability to generate new neurons from existing neural stem or progenitor cells. This process mainly occurs in specialized regions like the hippocampus and the subventricular zone (as mentioned earlier in the article). Research by Spalding et al. (2013) has shown that the human hippocampus retains its ability to generate neurons over a lifetime, which has implications for memory and mood regulation. By targeting the molecular pathways that regulate this process, such as the Wnt or Notch signaling pathways, we could potentially develop pharmaceutical interventions to enhance endogenous neurogenesis. These could be in the form of small molecules, peptides, or even gene therapies that aim to upregulate the body's natural capacity for neurogenesis.
However, the recruitment of new neurons through endogenous neurogenesis has some fundamental limitations. We must not only recruit from a neuron reservoir but also regulate the new neuron subtypes we wish to produce. This creates many challenges that may potentially be overcome through alternative strategies such as exogenous neurogenesis.
Exogenous Neurogenesis:
Exogenous neurogenesis in the context of this essay involves the production of new neurons outside of the body. These new neurons would need to be derived from multipotent cells and reincorporated into the brain as a cell-based therapy where they would finally integrate within the native neural circuitry. Such a bold strategy may have potentially limitless potential for augmenting our biological neural network. But is there any precedent for such an unconventional therapy?
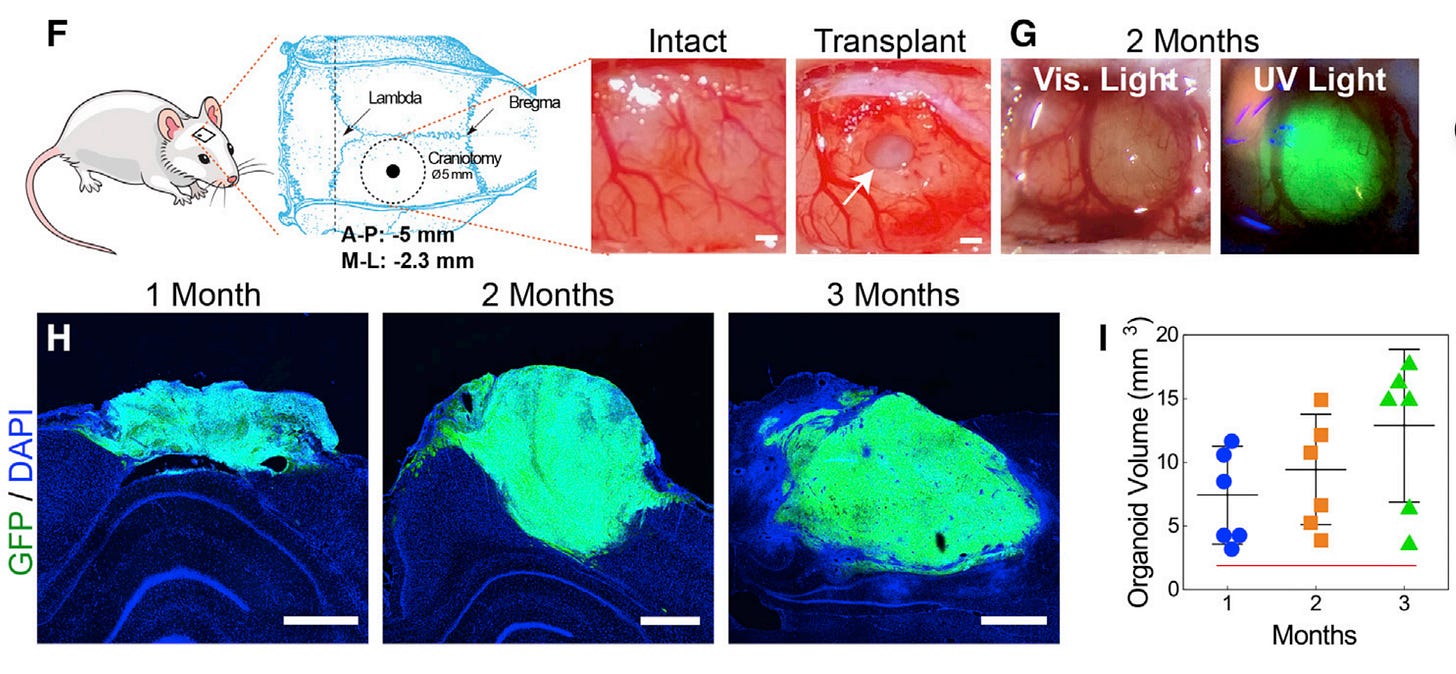
Recent research has been pushing the boundaries of what we know about neurogenesis and its potential applications. One groundbreaking study by Jgamadze et al. (2023) explored the structural and functional integration of human forebrain organoids with the injured adult rat visual system (Figure 3). These organoids are derived from skin cells that are transformed into stem cells through genetic reprogramming. This research is particularly relevant because it demonstrates the potential of human brain organoids to serve as a therapeutic strategy for restoring cortical function. The organoids were not only able to integrate structurally but also established synaptic connections with the host retina and visual system. Most impressively, these organoid neurons responded to host visual stimulation and even adopted feature selectivity. This suggests that we might be on the cusp of a new era where neurogenesis could be artificially induced and controlled to repair damaged neural networks.
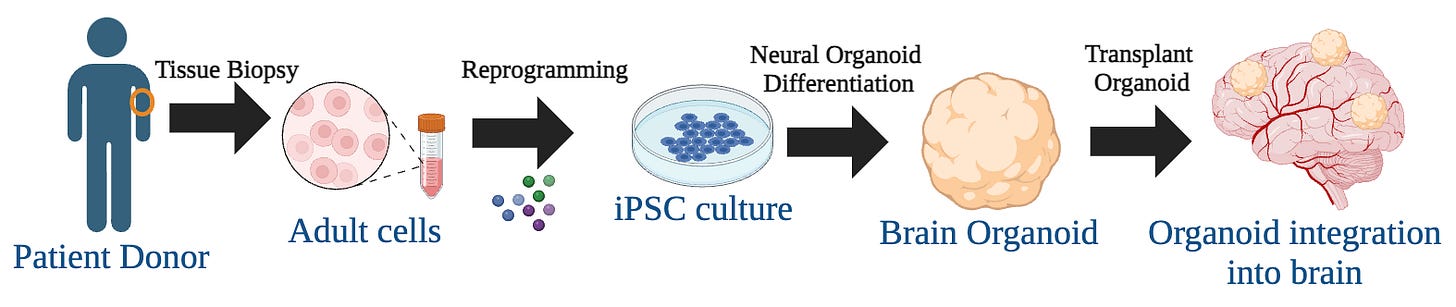
What would such a strategy look like for humans? I outline a potential strategy below (Figure 4).
Biopsy of patient fibroblasts: The first step involves extracting a skin biopsy from a patient. This is usually done using minimally invasive techniques. The overall goal is to retrieve fibroblasts that can be utilized for subsequent steps.
iPSC Induction: The extracted cells are then converted into induced pluripotent stem cells (iPSCs) using a process that involves the introduction of specific transcription factors (e.g., Oct4, Sox2, Klf4, and c-Myc) (Takahashi et al, 2007). These iPSCs can then be used to generate any cell type found in the body.
Cerebral Organoid Differentiation: Organoids are 3D cultures that mimic analogous tissues found within the body. Patient-derived iPSCs would then be guided to differentiate into brain organoids. Research by Lancaster et al, (2013) has shown that these organoids can mimic the cellular complexity of the human brain. More recently, researchers have demonstrated the generation of brain-region-specific organoids from human iPSCs (Qian et al, 2016).
Implantation: Finally, the organoid is transplanted back into the patient's brain. This is the most challenging step and involves precise surgical techniques to ensure that the new cells integrate properly without causing adverse effects. Some such evidence for the implantation of neurons has been demonstrated in humans: phase I clinical trial for treating Parkinson’s with dopaminergic neurons.
Gene Editing in vitro NPCs (Neuroprogenitor Cells):
Neuroprogenitor cells (NPCs) cultured in vitro provide an excellent platform for gene editing. Techniques like CRISPR-Cas9 could be used to edit genes that regulate neural development or function. For instance, a study from Bengoa-Vergniory et al explored the potential of CRISPR-Cas9 to correct mutations associated with neurodegenerative diseases (Bengoa-Vergniory et al, 2020). By editing the genes in NPCs before they are differentiated into neurons, we could potentially create neurons with enhanced capabilities or even correct genetic defects that lead to neurological diseases.
RISKS AND CHALLENGES
Now that we’ve explored the exciting possibilities of neurogenesis, let’s talk a bit about the inherent risks and challenges (at least the ones we can think of). Understanding these is key for future development of safe and effective therapies.
Endogenous neurogenesis in humans remains controversial. Some of the ongoing research has challenged the degree to which neurogenesis takes place in humans or even mammals in general. One such paper suggests that hippocampal neurogenesis drops sharply in children to undetectable levels in adults (Sorrells et al 2018). If this is true, then it will be important to know if NPCs still exist in the adult human brain. If not, exogenous neurogenesis may be the only option.
Overstimulation: Introducing new neurons into the brain might seem inherently beneficial, but there's a potential risk of overstimulation. The brain's neural networks have evolved to maintain a delicate balance. Excessive neuronal activity or density could disrupt this balance, potentially leading to neurological issues like increased seizure risk or sensory processing disorders or other unforeseen consequences. Furthermore, how many new neurons can we even fit safely into a human skull?
Integration Challenges: The native neural architecture is delicate. The addition of new neurons into this circuitry has potential to be harmful if it is done in a way that disrupts established circuits and communication. Therefore, therapeutic strategies would need to ensure the seamless integration of new neurons into existing neural circuits so that these neurons not only survive but also function harmoniously with existing networks. This is a tall order!
Long-term Effects: The long-term effects remain largely uncharted. Could there be a saturation point where additional neurons no longer provide benefits, or could they even start to impair brain function? These are questions that need thorough investigation.
CONCLUSIONS AND NEXT STEPS
As we navigate the intricate landscape of neurogenesis and its promising role in enhancing human intelligence, we stand at the cusp of a new era in both neuroscience and cognitive science. This exploration into the realms of endogenous and exogenous neurogenesis illuminates a path forward, where the augmentation of our biological capabilities might not just be a possibility but a reality.
Through our journey in understanding neurogenesis, from the intricate mechanisms that govern the birth of neurons to the potential cognitive enhancements they may bring, we have uncovered a world of opportunities tempered by significant challenges and ethical considerations. The prospect of enhancing memory, pattern recognition, and overall cognitive flexibility through neurogenesis presents an exciting frontier. However, this journey is also fraught with complexities, from the integration of new neurons into existing neural networks to the overarching implications of such interventions on the human experience.
As we advance, it is paramount that we proceed with a blend of scientific curiosity and ethical responsibility. The potential of neurogenesis to reshape our cognitive abilities and extend our intellectual horizons must be pursued with a keen awareness of the delicate balance within our neural ecosystems. Future research should not only focus on harnessing these biological processes but also on understanding their long-term implications, ensuring that our pursuit of cognitive enhancement aligns with the broader values and well-being of society. We again ask the question, could this and other such cognitive enhancement strategies result in additional emergent properties in humans?
The time is urgent. Throughout 2023, we have had constant and previously unfathomable innovation in the space of AI towards more and more human-like machines. We will rapidly find that we are outpaced by our machine counterparts as they continue to replace more and more of the cognitive labor space. It is important to understand our remaining runway for solving such a complex existential issue, which we will discuss in the next article.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
REFERENCES
Bengoa-Vergniory, N., Faggiani, E., Ramos-Gonzalez, P., Kirkiz, E., Connor-Robson, N., Brown, L.V., Siddique, I., Li, Z., Vingill, S., Cioroch, M. and Cavaliere, F., 2020. CLR01 protects dopaminergic neurons in vitro and in mouse models of Parkinson’s disease. Nature communications, 11(1), p.4885.
Boldrini, M., Fulmore, C. A., Tartt, A. N., Simeon, L. R., Pavlova, I., Poposka, V., ... & Hen, R. (2018). Human Hippocampal Neurogenesis Persists throughout Aging. Cell Stem Cell, 22(4), 589-599.
Clelland, C.D., Choi, M., Romberg, C.C.G.J., Clemenson Jr, G.D., Fragniere, A., Tyers, P., Jessberger, S., Saksida, L.M., Barker, R.A., Gage, F.H. and Bussey, T., 2009. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science, 325(5937), pp.210-213.
Faigle, R. and Song, H., 2013. Signaling mechanisms regulating adult neural stem cells and neurogenesis. Biochimica et Biophysica Acta (BBA)-General Subjects, 1830(2), pp.2435-2448.
Encinas, J.M., Michurina, T.V., Peunova, N., Park, J.H., Tordo, J., Peterson, D.A., Fishell, G., Koulakov, A. and Enikolopov, G., 2011. Division-coupled astrocytic differentiation and age-related depletion of neural stem cells in the adult hippocampus. Cell stem cell, 8(5), pp.566-579.
Jgamadze, D., Lim, J.T., Zhang, Z., Harary, P.M., Germi, J., Mensah-Brown, K., Adam, C.D., Mirzakhalili, E., Singh, S., Gu, J.B. and Blue, R., 2023. Structural and functional integration of human forebrain organoids with the injured adult rat visual system. Cell Stem Cell, 30(2), pp.137-152.
Lancaster, M.A., Renner, M., Martin, C.A., Wenzel, D., Bicknell, L.S., Hurles, M.E., Homfray, T., Penninger, J.M., Jackson, A.P. and Knoblich, J.A., 2013. Cerebral organoids model human brain development and microcephaly. Nature, 501(7467), pp.373-379.
Mu, Y., Lee, S.W. and Gage, F.H., 2010. Signaling in adult neurogenesis. Current opinion in neurobiology, 20(4), pp.416-423.
Okano, H., Temple, S. (2009). Cell types to order: temporal specification of CNS stem cells. Current Opinion in Neurobiology, 32, 84–92.
Toni, N., Teng, E.M., Bushong, E.A., Aimone, J.B., Zhao, C., Consiglio, A., van Praag, H., Martone, M.E., Ellisman, M.H. and Gage, F.H., 2007. Synapse formation on neurons born in the adult hippocampus. Nature neuroscience, 10(6), pp.727-734.
Sahay, A., Scobie, K.N., Hill, A.S., O'Carroll, C.M., Kheirbek, M.A., Burghardt, N.S., Fenton, A.A., Dranovsky, A. and Hen, R., 2011. Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature, 472(7344), pp.466-470.
Sorrells, S.F., Paredes, M.F., Cebrian-Silla, A., Sandoval, K., Qi, D., Kelley, K.W., James, D., Mayer, S., Chang, J., Auguste, K.I. and Chang, E.F., 2018. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature, 555(7696), pp.377-381.
Takahashi, K., Tanabe, K., Ohnuki, M., Narita, M., Ichisaka, T., Tomoda, K. and Yamanaka, S., 2007. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. cell, 131(5), pp.861-872.
Vaz, A., Ribeiro, I. and Pinto, L., 2022. Frontiers in Neurogenesis. Cells, 11(22), p.3567.



Have we tried neurogenesis on animals yet? I'm really curious to see if there's any crossover or studies we could do before we try humans!
Exogenous neurogenesis! Wow, this is super interesting. I did not consider exogenous methods in my future Neurobionics fiction scenario where targeted neuroplasticity results in certain types of humans more apt for certain purposes than others, combined with BMI tech to solve the "space" issue, in my fiction that is. As you say, how much can you cram in that head?
"These new neurons would need to be derived from multipotent cells and reincorporated into the brain as a cell-based therapy where they would finally integrate within the native neural circuitry."
Integration is the hardest part? Rejection rates, infection, tissue deterioration, precision? I can't begin to wonder about long-term effects, either. Regardless, it's going to happen. Isn't it?