Top 5 Biotech Breakthroughs that shaped 2025
Reflecting on Biotechnology this past year
Welcome to 2026, Readers! Let’s stay relentless this year!
I’m excited to kick off the year and share the Top 5 Biotech Breakthroughs of 2025. This continues a tradition I began in 2023, highlighting what I believe are the most transformative technologies and innovations that shape the biotech landscape over the past 12 months. This exercise is helpful to me as a scientist and writer, since it gives me time to reflect on the progress and understand how these advancements will shape the future of healthcare, longevity, and beyond.
This year’s review has its own twist. Rather than assembling the list alone, I invited four exceptional writers and thinkers in the biotech space to each contribute what they believe was the most important biotech breakthrough of 2025. The result is a set of five breakthroughs that reflect not a single perspective, but a convergence of independent judgment across the field. I strongly encourage you to check out the guest contributors shown below, each has a distinct voice and set of interests that’s well worth following.
List
Obesity has a Pharmacological Solution. Body Composition is next.
AI drug development: protein design for multi-step enzymes
The Fully Automated Laboratory
Alzheimer’s & Dementia Progress
Cell Therapy Advancements.
Guest authors:
Stephen Turner - Paired Ends
Owen Lewis - Techno-Optimist Substack
Riccardo Scribano - BioDodo
Tony Huge - Tonyhuge.is
With that, let’s dive in.
First, if you enjoy these posts, consider subscribing and becoming a part of our growing community!
Obesity has a Pharmacological Solution. Body Composition is next.
David Kingsley & Tony Huge
Obesity is not only an aesthetic issue. It is a chronic disease and inflammatory state that raises risk for hypertension, dyslipidemia, type 2 diabetes, heart disease and stroke, multiple cancers, chronic kidney disease, and fatty liver disease (CDC). Creating a biotech solution to obesity will have a huge improvement in the healthspan and lifespan we see in our society, as well as impact overall healthcare costs and usage. The good news is that we are getting close.
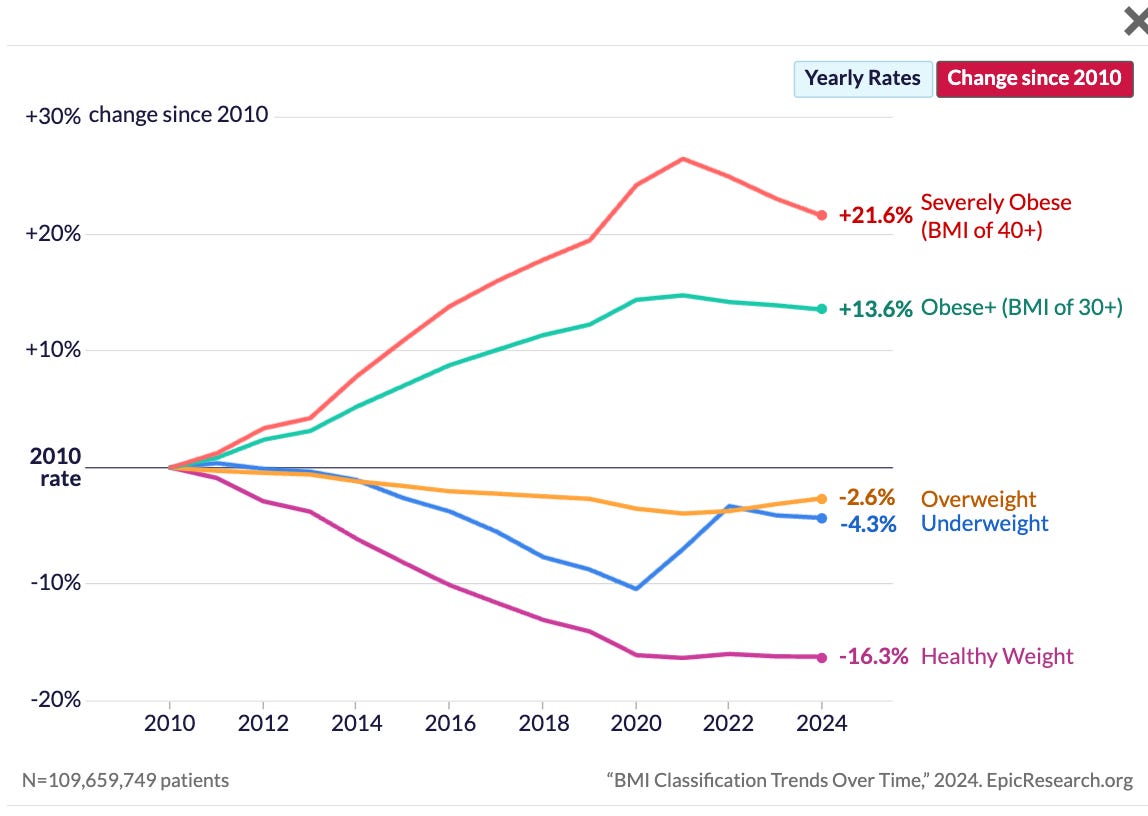
This year marked another significant leap towards a pharmacological solution to obesity, where drug efficacy rivals bariatric surgery. How did we get there and what are the results?
Incretin-mimetic drugs emerged as a breakthrough for weight management in the early 2020s, led by the repurposed diabetic agent semaglutide. As a GLP-1 agonist, semaglutide demonstrated that double-digit weight loss (~15% vs. placebo) was achievable (Rubino et al., 2022). These compounds function by mimicking natural gut hormones, triggering a cascade of downstream effects including optimized insulin release, blood glucose regulation, delayed gastric emptying, and appetite suppression. This success paved the way for next-generation agents like tirzepatide (a dual GLP-1/GIP agonist), which pushed weight reduction upward of 20% over 72 weeks. Yet, these drugs represent only the beginning; significant progress over the past year has built upon this foundation to further enhance efficacy and optimize body composition.
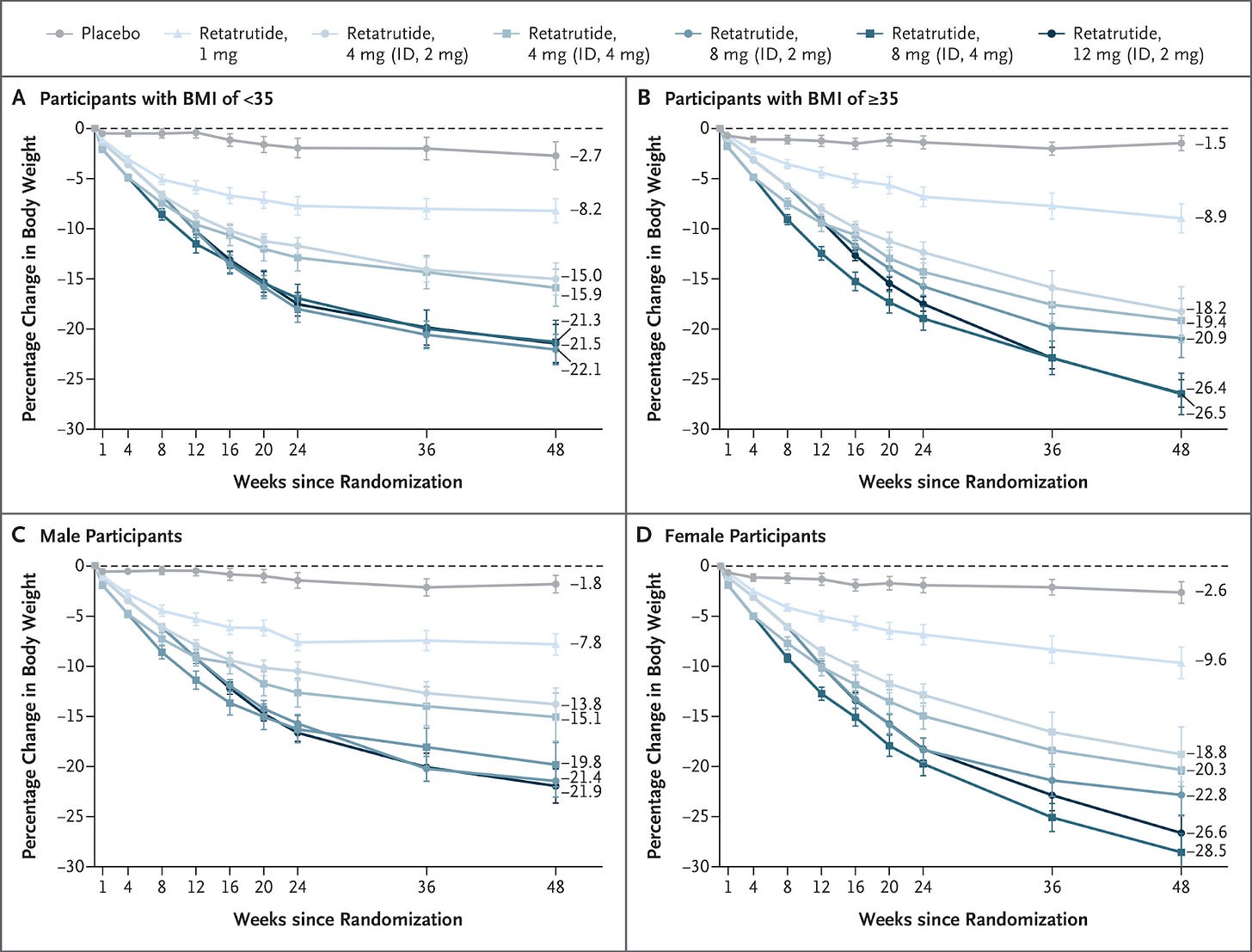
Multi-agonist therapies and combinations led the charge. The newest wave of incretin-based drugs and dosing procedures now consistently produces ≥20% body weight loss, a threshold once unimaginable for medications. In the SURMOUNT trials, tirzepatide (now FDA-approved as Zepbound) achieved roughly 18–22% average weight reduction over 72 weeks (Jastreboff et al., 2022). The following year, Lilly’s retatrutide pushed this further with Phase 2 results showing 24.2% mean weight loss at 48 weeks on the highest dose, with patients losing an average of 58 pounds and not yet reaching a plateau by study end (Jastreboff et al. 2023). In December, Lilly shared results from retatrutide’s Phase 3 TRIUMPH-4 study showing an average 28.7% mean weight loss over 68 weeks alongside significant reductions in osteoarthritis-related knee pain, with more clinical endpoints to be released in 2026. This is huge!
Changes in Body Weight with Retatrutide as Compared with Placebo (Jastreboff et al. 2023).
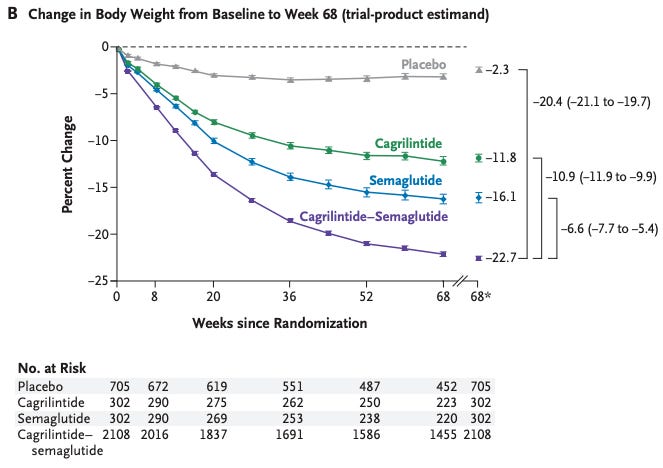
Likewise, Novo Nordisk’s combo CagriSema (cagrilintide + semaglutide) demonstrated a 22.7% mean weight reduction at 68 weeks in a recent trial (Garvey et al., 2025). Over 40% of CagriSema-treated patients dropped at least a quarter of their body weight, a degree of efficacy that would have been science fiction a few years ago. These advances firmly establish 20%+ weight loss as the new standard for anti-obesity drugs, in contrast to the ~5–10% from older therapies.
Change in Body Weight with Cagrilintide–Semaglutide as Compared with Semaglutide, Cagrilintide, and Placebo (Garvey et al., 2025).
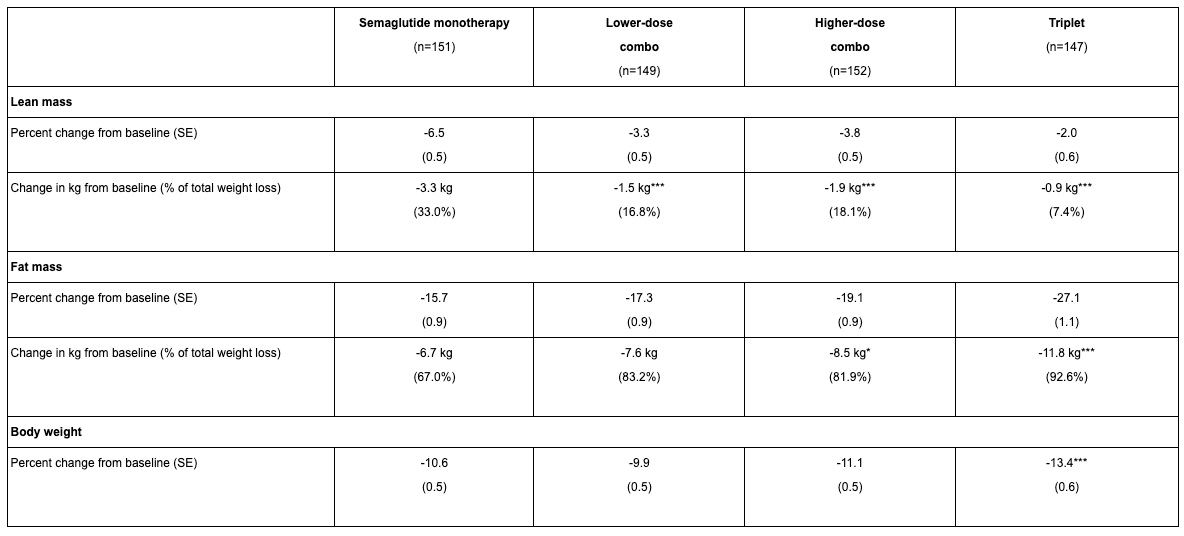
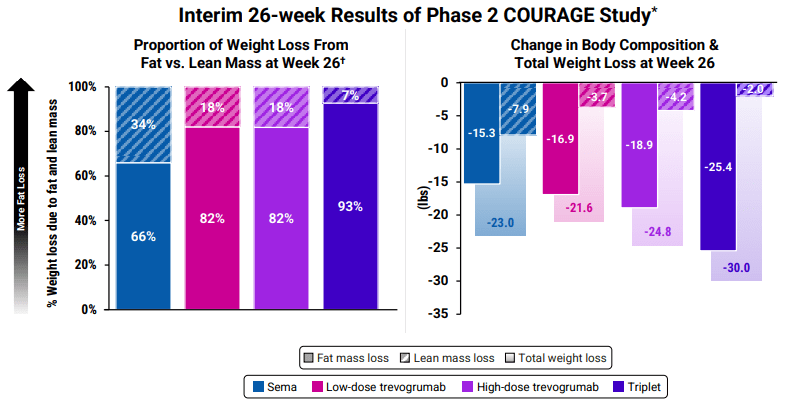
Body composition is now a focus by including muscle-preserving adjuvant therapeutics. Equally important, the field is shifting focus from weight loss alone to body composition engineering. It’s no longer just about “how much” weight is lost, but “what” is lost, i.e., fat vs. lean mass. Potent incretin mimetics like GLP-1 agonists inherently cause both fat and muscle loss, so researchers are now targeting fat selectively. A landmark Phase 2 study (the BELIEVE trial) shared at American Diabetes Association combined semaglutide with bimagrumab, a myostatin/activin pathway antibody designed to spur muscle growth, and saw striking results. On semaglutide alone, users lost about 15.7% body weight with about 72% of each pound shed being fat tissue, with the rest being lean tissue loss. With the addition of bimagrumab, total weight loss was 22.1% with over 92% of weight loss from fat. Even more interesting, in bimagrumab alone, 100% of weight loss was attributed to fat mass and there was a modest increase in lean muscle (~2.5%). This approach yielded dramatic improvements in waist circumference and other metabolic markers by preserving muscle. Lilly has since launched their own study pairing acquired Bimagrumab with their flagship drug Tirzepatide.
Similar results are being observed in Regeneron’s Courage Trial, a 26-week study combining semaglutide with Trevogrumab (anti-GDF8/myostatin) and/or Garetosmab (anti-activin A) for treating obesity (Regeneron Press). Early results reflect what is seen with Bimagrumab and Semiglutide, i.e., maintain more lean muscle mass and lose additional fat mass. However, even without adjunct therapy, newer drugs are being evaluated for their impact on body composition. For example, retatrutide’s trials included a body composition substudy that showed minimal lean mass loss despite massive fat reductions. The emphasis on “quality” of weight loss, maximizing fat reduction while maintaining muscle, is now front and center in obesity pharmacology.
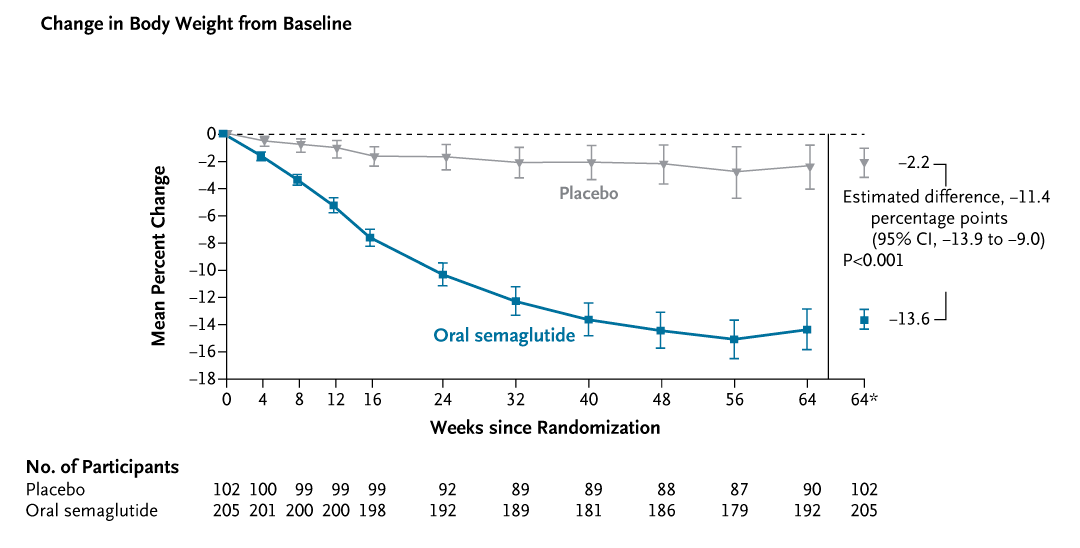
Formulation and convenience being tackled. Another breakthrough of 2025 was the success of oral formulations of these typically injectable peptides. Novo Nordisk presented high-dose oral semaglutide tablets (25 mg and 50 mg daily) that match injection efficacy. In the Phase 3 OASIS 4 trial, oral semaglutide 25 mg produced a 14% mean weight loss at 64 weeks versus 2% for placebo, essentially the same benefit as the 2.4 mg weekly injection (Wharton et al., 2025). Notably, 30% of patients on oral semaglutide achieved ≥20% weight loss (vs only 3% on placebo). These results were on par with the 50 mg dose and confirm that pill-based GLP-1 therapy can deliver surgery-level weight reductions.
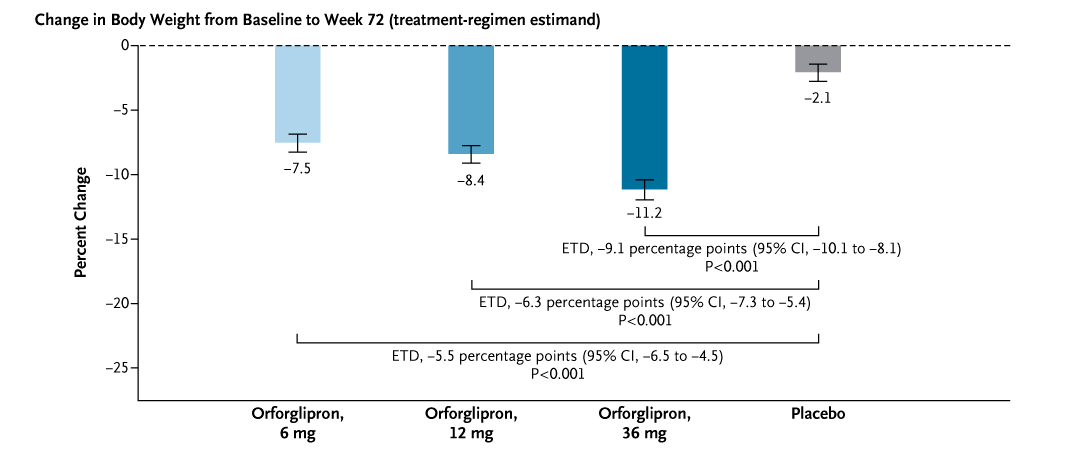
Meanwhile, Eli Lilly’s orforglipron, the first non-peptide (small-molecule) GLP-1 receptor agonist in pill form also made headlines. Orforglipron phase 3 (ATTAIN‑1) showed dose‑dependent weight reduction at 72 weeks (highest dose ~−11.2% vs −2.1% placebo), with “nearly one fifth” of participants at the highest dose reaching ≥20% weight loss; adverse events were predominantly gastrointestinal and usually mild‑to‑moderate (Wharton et al., 2025).
Although injectables still have an edge in absolute potency, oral options are rapidly closing the gap. The convenience of pills (no needles or cold storage) could hugely expand access, making long-term therapy more feasible for millions.
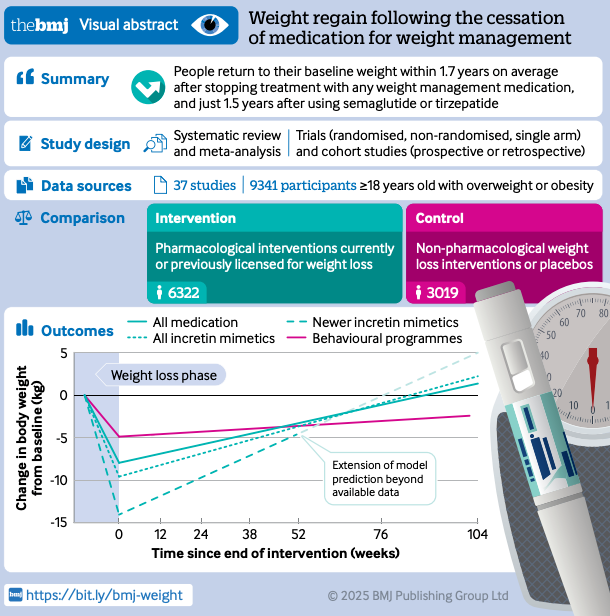
Future challenges remain with tolerability and weight regain after cessation. Despite remarkable efficacy, safety and tolerability remain key bottlenecks. All these agents work via potent hormonal effects on appetite and digestion, so gastrointestinal side effects are ubiquitous. Nausea, vomiting, and diarrhea are by far the most common adverse events and tend to occur at higher doses. For example, in trials of the GLP-1 combo CagriSema, about 80% of patients experienced some GI symptoms (mostly mild-to-moderate) compared to ~40% on placebo (interesting placebo is 40%). Typically 5–10% of participants cannot tolerate the full dose and discontinue treatment due to side effects. Additionally, class-wide safety considerations (such as rare risks of pancreatitis, gallbladder complications, or medullary thyroid cancer in predisposed patients) necessitate ongoing vigilance. So far, no new serious hazards have emerged in the latest trials – adverse events have remained in line with known GLP-1 class effects and generally manageable. Nevertheless, improving the side effect profile is a priority. Approaches in development include better targeting of receptors, alternative hormones (e.g. amylin agonists with fewer GI effects), and adjunctive therapies (like bimagrumab) to allow lower dosing of incretins. In short, the efficacy is here, but making treatment tolerable for all is the next hurdle. Perhaps the single biggest draw back for users of incretin products is that people seem to regain the majority of weight within 2 years of cessation of these products. So, great news for the pharma companies and their investors, not so great for users (West et al., 2025).
AI drug development: protein design for multi-step enzymes
Stephen Turner - Paired Ends
A useful way to frame 2025’s AI drug development progress is that we are no longer just predicting structures or ranking candidates. We are starting to design and construct new functional biomolecules with performance that has to survive contact with reality: purification, kinetics, and crystal structures.
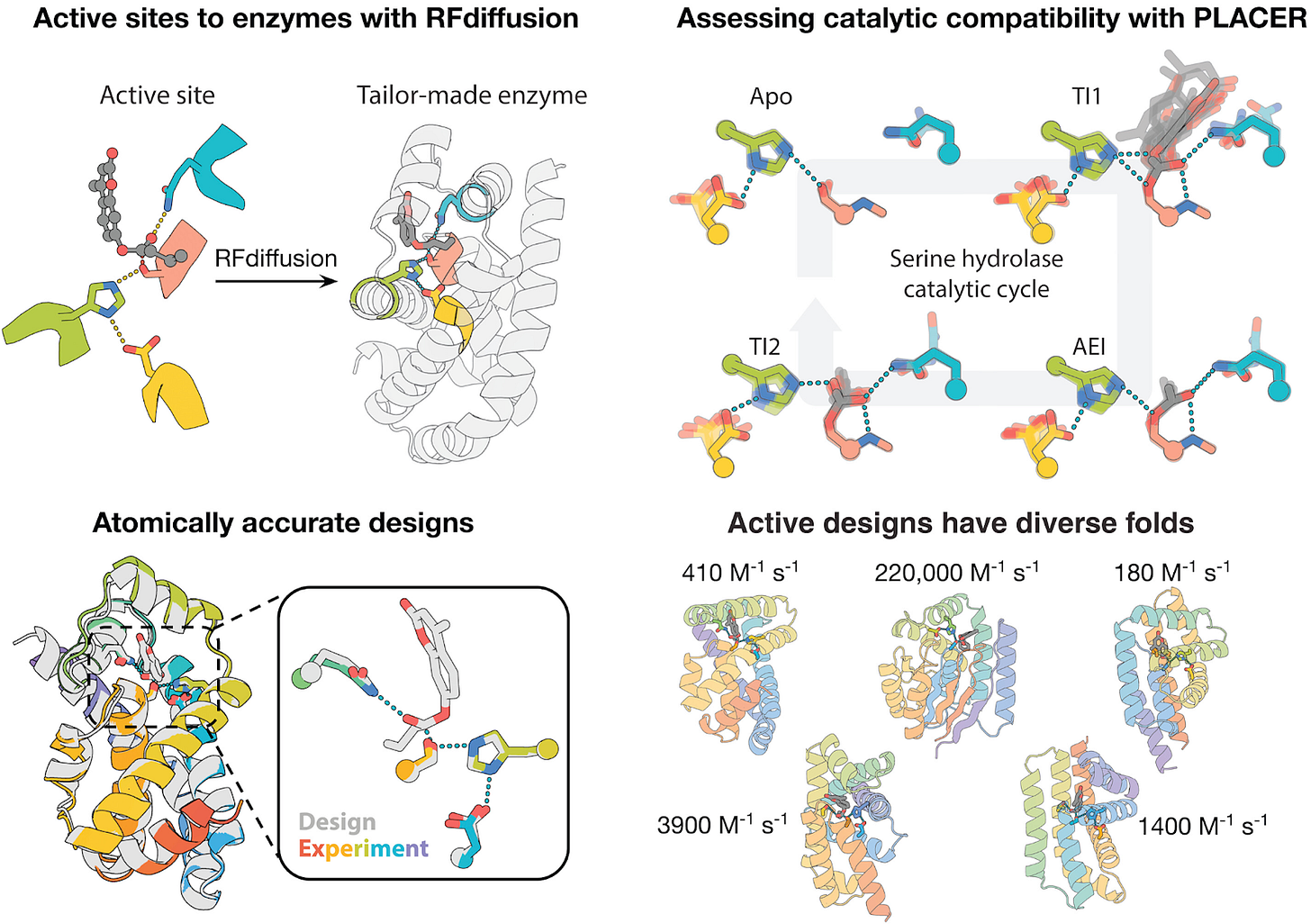
A striking recent example comes from David Baker’s group, who used a generative protein design workflow centered on RFdiffusion and then used AlphaFold2 as an in silico gatekeeper before committing designs to the wet lab.
Lauko, Anna, et al. “Computational design of serine hydrolases.” Science 388.6744 (2025): eadu2454. https://www.science.org/doi/10.1126/science.adu2454
In this work they explicitly set out to tackle a multistep catalytic cycle, not a single transition state. That matters because multistep enzymes must stabilize and choreograph multiple transition states and intermediates, including an acyl-enzyme intermediate and two tetrahedral intermediates, with tight geometric preorganization at each step.
Methodologically, the paper is a blueprint for how AI components are starting to compose: RFdiffusion is used to generate backbones around a target catalytic geometry; sequence is designed with LigandMPNN and refined; AlphaFold2 is used to filter for designs whose catalytic residues land within 1A of the intended configuration. They then add a key twist: PLACER, an ensemble modeling approach, to evaluate compatibility and preorganization across the entire reaction coordinate, which improves success rates and activity.
This work is not incremental. They report multiple designed serine hydrolases with crystal structures that match the design models at sub-angstrom accuracy and extremely high catalytic efficiencies. Even more interesting, the best examples appear to extend beyond known hydrolase fold space: Foldseek finds only weak structural matches (TM scores near or below 0.5), and the closest hits are proteins of unknown function with no similarity to known hydrolases at fold or active-site level.
Zooming out, this is why AI drug development hits different now. It is becoming a general capability: specify function, generate matter, validate, iterate, and eventually industrialize.
RFdiffusion generates enzymes tailor-made for a given active site (top left). Assessing design compatibility across the catalytic cycle with the PLACER neural net. Conformational ensembles for each step of the reaction are shown (top right). Comparison with crystal structures shows that designs made with RFdiffusion and filtered with PLACER are atomically accurate (bottom left). Designed serine hydrolases expand the fold space of this ancient enzyme family (bottom right, catalytic efficiencies listed).
The Fully Automated Laboratory
Stephen Turner - Paired Ends
David Kingsley - Neural Nexus
LLMs and Advanced robotics will make laboratories autonomous. We are seeing the first steps towards this. The core idea is not “a chatbot that writes protocols.” It is closing the loop between hypothesis, execution, measurement, and iteration, with software that can reason over experiments the way a strong scientist does, while operating at machine cadence.
For the first time, an AI system demonstrated it could autonomously design and optimize real-world experiments, essentially acting as a scientific “control plane” in the laboratory. OpenAI’s wet-lab evaluation is a concrete early signal:
OpenAI Research Blog: Measuring AI’s capability to accelerate biological research in the wet lab (Dec 16, 2025).
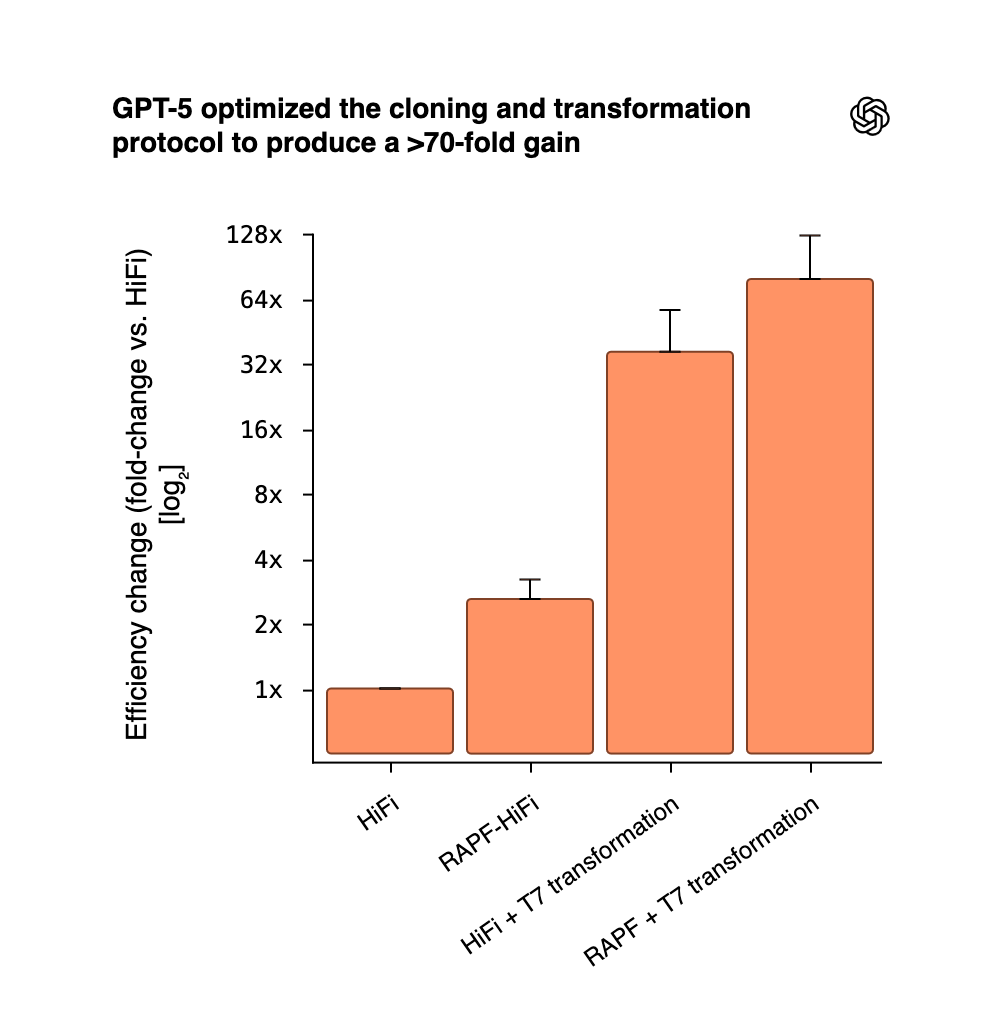
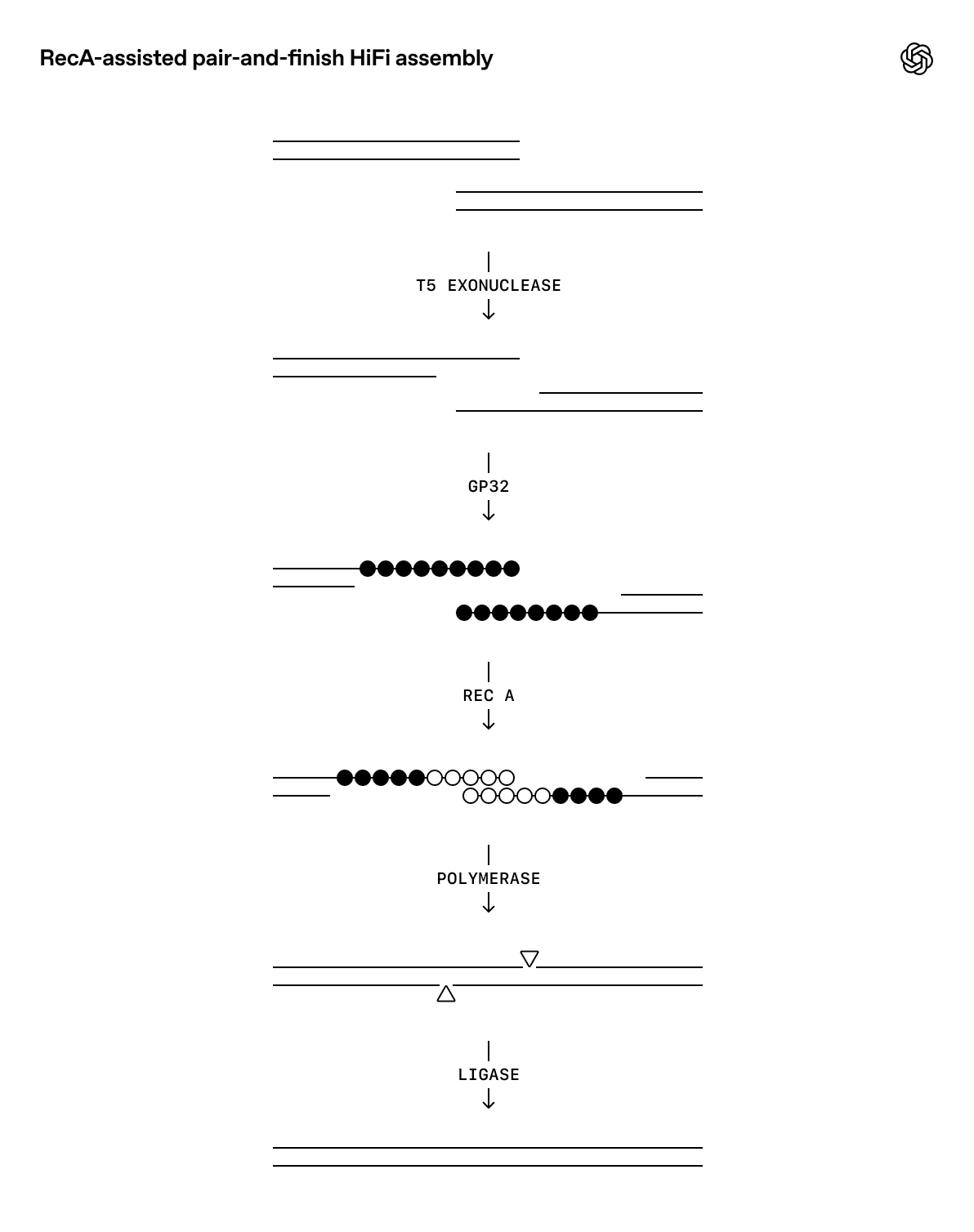
The landmark example came from OpenAI’s collaboration with biosecurity startup Red Queen Bio: using GPT‑5, the team built a closed-loop framework where the LLM proposed experimental protocols that human scientists executed, feeding results back to the model. Through iterative refinement, GPT‑5 optimized a standard molecular cloning procedure by a factor of 79 – an unprecedented gain. Notably, the model’s suggestions were not simply copied from literature; it introduced new elements (like adding E. coli RecA recombinase and a phage DNA-binding protein to the DNA assembly process) that hadn’t been used in that protocol, leading to dramatically higher yields. Researchers observed that GPT‑5 integrated known biological mechanisms in a novel way, showing “glimpses of creativity” beyond what its training data alone would predict. While not a fundamental discovery in biology, this achievement proved that an LLM can drive experimental innovation in the lab, not just analyze text
Autonomous lab robotics played a key role in extending the LLM’s control to physical execution. In the GPT‑5 cloning experiment, Red Queen Bio and Robot.dev built a custom robotic system that could carry out GPT‑5’s instructions in the lab via natural language commands. The setup included a human-to-robot LLM interface (to translate plain English protocols into robot actions), a vision module for real-time labware identification, and a motion planner for precise liquid-handling and incubation steps. In trials, this autonomous robot handled complex biotech procedures – mixing reagents, heat-shocking cells, plating cultures – and reproduced the improved cloning protocol almost as effectively as a human scientist. The robot-achieved outcome was ~89% of the human-run experiment’s yield, demonstrating that LLM-directed experiments can be automated end-to-end with only minimal performance loss so far. These results point toward a future where LLM “agents” not only design experiments but also directly orchestrate laboratory robots to execute them.
Underpinning this progress is the maturation of cloud laboratories, a highly automated lab facilities accessible via software APIs. Platforms like Emerald Cloud Lab (ECL) and Strateos reached new levels of adoption by 2025, allowing researchers (or their AI assistants) to remotely run experiments on dozens of instruments through a web interface. In fact, Carnegie Mellon University opened the world’s first academic cloud lab (in partnership with ECL) in early 2024, housing over 200 robotic instruments available on demand. This “self-driving lab” at CMU’s Bakery Square facility enables scientists to submit experiment instructions digitally and get back results without ever entering a physical lab. Early integrations with LLMs hint at how powerful this model can be: researchers have prototyped LLM-based lab assistants that take a natural language experiment description and translate it into cloud lab protocols automatically. In other words, a scientist could soon “chat” with an AI to run an assay, with the LLM handling all the low-level method details and instrument control. Such systems remain in beta, but the concept has been proven in principle by projects like CMU’s Coscientist, which linked a GPT-4 agent to ECL’s robotic platform to autonomously carry out chemistry reactions based on a plain-English goal. The democratizing effect is significant – by abstracting lab work into software, these AI-driven cloud labs can make advanced experimentation accessible well beyond elite research centers.
Major industry players also pushed wet-lab AI integration in 2025. In February, Google unveiled an “AI co-scientist” designed to act as a virtual collaborator for biomedical research. The system uses a powerful LLM (from Google DeepMind’s Gemini family) to absorb vast scientific literature and generate hypotheses or experiment plans for scientists. In a proof-of-concept study on liver fibrosis, Google reported that the AI co-scientist suggested several novel therapeutic approaches – and when these suggestions were tested in vitro by human researchers, all of them showed promising activity against the disease mechanism. This was a striking validation of LLM-generated hypotheses in a real biomedical context. Later in the year, DeepMind announced it will open an automated lab in the UK in 2026 as part of a government partnership. That facility is being built specifically to integrate DeepMind’s multimodal Gemini LLM with robotics, enabling high-throughput experiments in materials science (synthesizing and testing hundreds of samples per day) with minimal human intervention. Together, these efforts by tech leaders show a clear trend: LLMs are moving from the computer room into the laboratory, becoming central to how experiments are conceived and conducted.
Beyond these headline projects, 2025 also saw LLMs permeating everyday scientific workflows. Many labs began using LLM “copilots” for routine tasks like analyzing data, reading logs, and drafting experiment plans. For instance, electronic lab notebook platforms (e.g. Benchling) introduced GPT-powered features that let scientists query their experimental records in natural language and get summaries or insights instantly. LLMs showed proficiency at interpreting unstructured notes or instrument outputs and suggesting next steps – effectively acting as intelligent lab assistants. In one notable case, researchers developed a specialized agent dubbed “CRISPR-GPT” that was fine-tuned on gene editing knowledge and linked to lab automation tools. This system autonomously designed CRISPR guide RNAs, planned the gene knockout/activation experiments, and analyzed the results, successfully executing several genome edits in human cells with no human design input. Meanwhile, IBM researchers demonstrated ChemCrow, an LLM-based chemistry assistant that uses GPT-4 plus domain tools to plan and carry out chemical syntheses – it even proposed and helped create a new fluorescent molecule that standard methods hadn’t found. And at Carnegie Mellon, the Coscientist system proved capable of handling entire reaction workflows: given a target outcome, it retrieved relevant literature, wrote Python code to control liquid-handling robots, ran the experiment in a cloud lab, and iteratively refined conditions based on the data. These real-world examples underscore that LLMs can go well beyond writing reports – they are now generating hypotheses, planning experiments, and controlling lab hardware in various scientific domains.
Humanoid robots emerged as practical wet-lab automatons in 2025 by virtue of their human-mimicking form, which lets them function with existing benches, pipettes, centrifuges, and other tools built for human ergonomics. Unlike fixed automation, a bipedal robot can navigate legacy lab infrastructure and operate standard instruments without costly reconfiguration. Crucially, several high-profile pilots demonstrated this advantage: a biotech R&D lab deployed a humanoid “scientist” that performs pipetting and reagent handling tasks on human-standard equipment; Figure AI partnered with BMW to trial humanoids on car assembly lines (moving the technology from lab demos to real production); Apptronik’s Apollo robot was piloted in Mercedes-Benz facilities to deliver parts and inspect components in human-designed workspaces; and Tesla began using Optimus prototypes in its own factories and engineering labs for basic material-handling chores. Tightly integrating these humaniform actuators with an LLM-driven “control plane” means an AI can devise experimental plans and have the robots execute them end-to-end – effectively serving as the hands of an autonomous lab that accelerates experimentation in environments originally built for human scientists.
Alzheimer’s & Dementia Progress
Owen Lewis - Techno-Optimist Substack
Anyone who has watched a loved one suffer from Alzheimer’s or dementia knows the frustration and hopelessness the diagnosis brings. While three monoclonal antibodies, Aducanumab, Lecanemab, and most recently Donanemab, have now been approved to slow early Alzheimer’s, even the best of them reduces progression by only about 35% and comes with serious risks like brain bleeds. But I think 2025 marks the beginning of a real shift. Over the past year, a steady tempo of breakthroughs is painting a different picture. Most won’t help patients immediately, but the progress of the past year has us on a path where Alzheimer’s and dementia could be more treatable, or even curable, within the next decade.
We’re starting to uncover the root causes of Alzheimer’s and dementia, with scientists finding a direct cause and effect relationship between malfunctioning mitochondria, the energy-producing “engines” of cells, and memory loss seen in these neurodegenerative diseases. Ramping up mitochondrial activity in mice restored memory performance. Another insight might have uncovered a more common genetic cause, with a protein called PHGDH potentially disrupting how brain cells regulate genes, which can trigger disease onset. They also identified a small molecule that blocks this harmful activity without interfering with the gene’s normal function. In mouse models, it slowed disease progression and improved memory.
Quick intervention will always be best, with a molecule called WIN55.212-2 found to protect the brain and restore cognitive function in early Alzheimer’s. Rodent tests have been very promising, though a problem here is the molecule is “free to use,” meaning it cannot be patented as a drug—decreasing the likelihood a pharmaceutical company would move forward with it.
Some treatments don’t require drugs at all, with the use of low-intensity ultrasound on the brains of mice with an Alzheimer’s disease like condition boosting cognitive function, though it didn’t clear amyloid plaques. Early human trials have been done by the team, but annoyingly the article didn’t say what (if any) improvements were seen. The impression I get though is that it was effective.
Turning to the periodic table, a study found that the noble gas Xenon could treat Alzheimer’s disease. Inhaling the gas “reduced neuroinflammation, minimized brain atrophy, and promoted protective neuronal states in mouse models of Alzheimer’s disease.” Human trials have (I think) already started. Harvard researchers have discovered that lithium naturally plays a crucial role in maintaining neural health—and that its depletion appears to be one of the earliest detectable signs of Alzheimer’s. In mice, lowering brain lithium accelerated damage, including amyloid plaques, tau tangles, inflammation, and memory loss, while a lithium compound (lithium orotate) that avoids binding with plaques reversed those effects and restored cognitive function. The best part is that lithium orotate is already an approved treatment for other things and is considered safe.
Another report found that the common amino acid arginine can reduce Alzheimer’s-like brain damage in animal models by inhibiting toxic amyloid aggregations while also dampening neuroinflammation. Oral arginine improved cognitive performance and slowed disease progression, suggesting benefits beyond plaque reduction alone. Because arginine is already widely used in humans with a strong safety profile, the findings point to a potentially low cost, fast intervention if the effects hold up in clinical trials.
Nanotechnology could play a role as well, with engineered nanoparticles slashed amyloid-beta levels by 50-60% within one hour in Alzheimer’s mice by reactivating the blood-brain barrier’s natural waste removal system, rather than directly attacking the proteins. Cognitive improvements lasted 6 months, a significant chunk of a mouse’s lifespan.
A rather unexpected finding is that the shingles vaccination may slow dementia progression, not just reduce risk. Researchers found vaccination was associated with less cognitive impairment in healthy people, lower conversion from Mild Cognitive Impairment to dementia, and reduced mortality in those already diagnosed, across Alzheimer’s, vascular dementia, and most strongly, mixed dementia. The effect has now been replicated multiple times, with randomized trials underway. Serious message here, everyone tell your parents and grandparents to get this. Get it yourself if you’re 50+.
I’ve saved the best for last: Researchers have reversed advanced Alzheimer’s disease in mice by restoring the brain’s energy balance through a molecule called NAD+, which naturally declines with age and drops even more severely in Alzheimer’s patients. Mouse models showed complete cognitive recovery even after the disease was already advanced. The treated mice showed normalized blood biomarkers for Alzheimer’s, and complete recovery of all cognitive functions. The key takeaway is a message of hope: the effects of Alzheimer’s disease may not be inevitably permanent. The damaged brain can, under some conditions, repair itself and regain function.
Cell Therapy Advancements
Riccardo Scribano - BioDodo
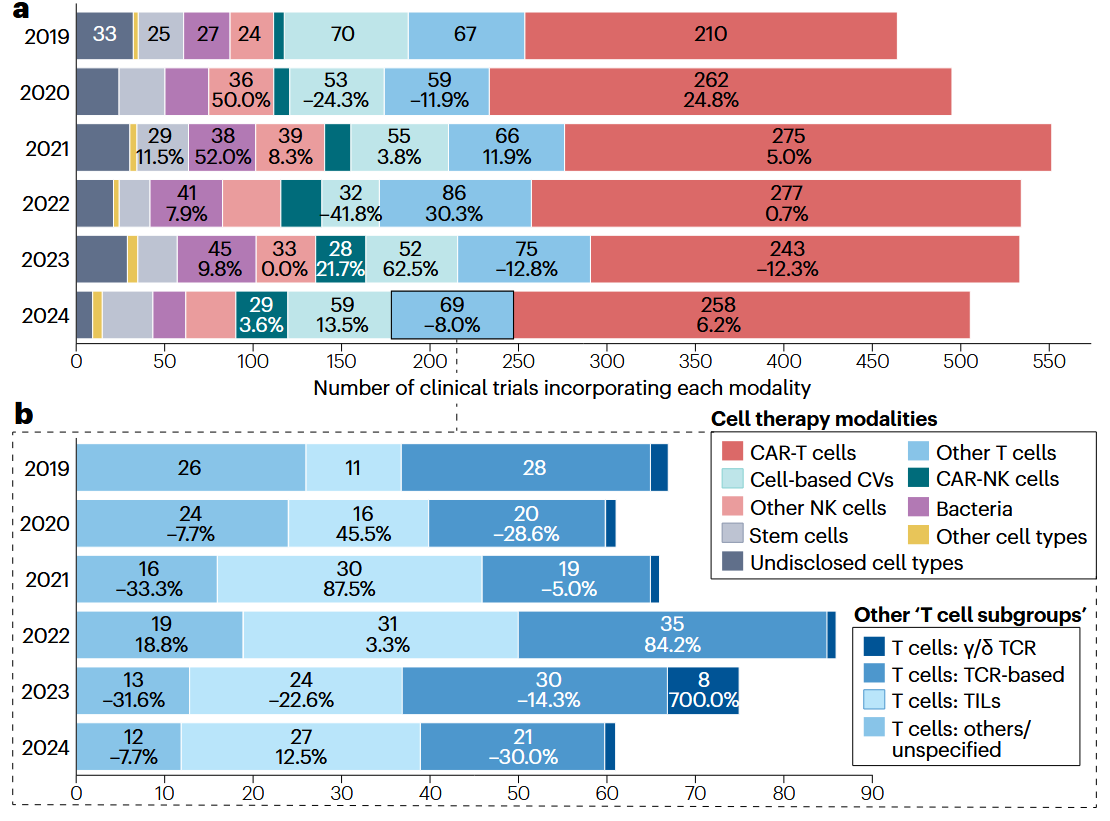
Since the first FDA approval of the first Chimeric antigen receptor engineered T-cells (CAR-T) in 2017, cell therapies have transformed the treatment of hematological malignancies, delivering high response rates while revealing limitations (Shokati et al., 2025). By mid-2025, the field has expanded to more than 6,000 registered interventional cell therapy trials worldwide, with CAR-T still dominating the landscape. Hematological cancers still represent the largest category but are steadily contracting, alongside sustained industry interest and diversification of targets and platforms (Benthani et al., 2025).
Source: Benthani et al., 2025
A major focus of recent progress lies in solid tumors, which remain challenging because of poor CAR-T trafficking and infiltration, physical and immunosuppressive tumor microenvironments, and antigen heterogeneity leading to immune escape. Nonetheless, encouraging signs have emerged from clinics in 2025 (Escobar et al., 2025). A phase-1 clinical trial on intrinsic pontine glioma, an aggressive brainstem tumor, has shown the safety of repeated intracerebroventricular CAR-T treatment (Vitanza et al., 2025). CAR-macrophages (CAR-M) have gained momentum due to their innate capacity to infiltrate solid tumors, with early-phase trials demonstrating favorable safety profiles, tumor microenvironment remodeling and disease stabilization in HER2-positive cancers (Abdin et al., 2024; Reiss et al., 2025). Still, the only FDA-approved cell therapies for solid tumors remain Lifileucel (Amtagvi), derived from tumor-infiltrating lymphocytes (TIL) and used to treat melanoma, and Afamitresgene (Tecelra), an engineered T-cell receptor (TCR) therapy against synovial sarcoma.
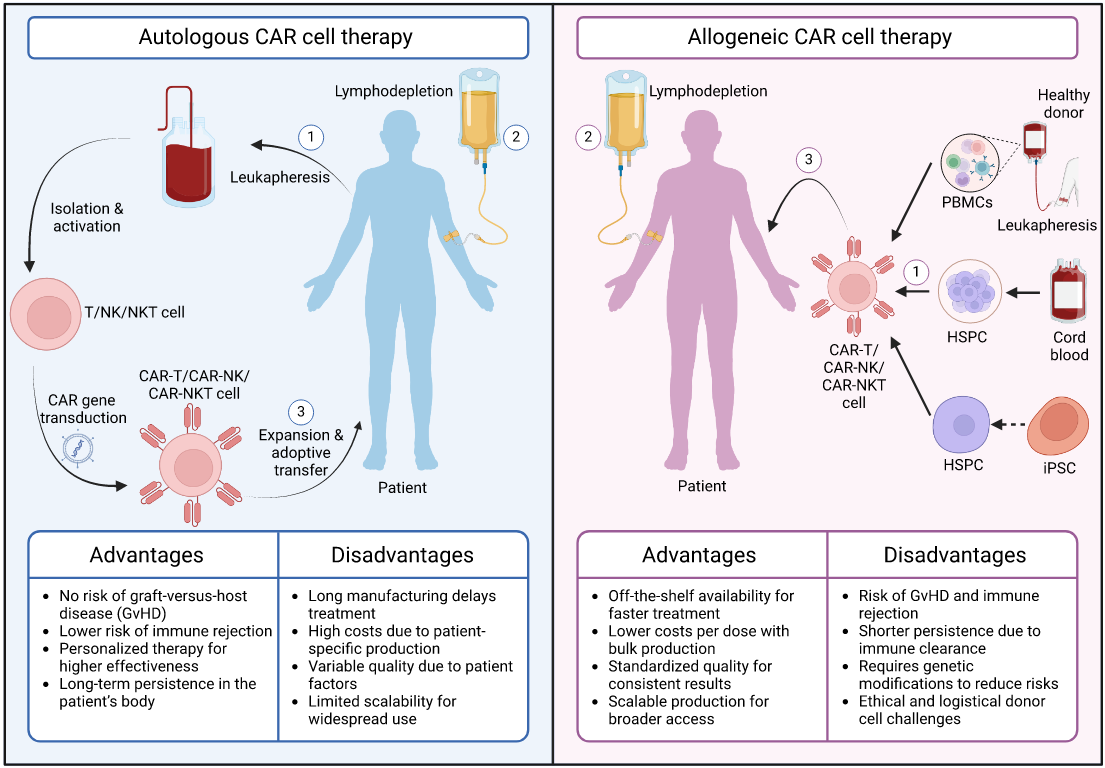
In parallel, allogeneic “off-the-shelf” CAR-T and CAR-Natural killer (CAR-NK) programs have stabilized and expanded, leveraging healthy donor- or stem-cell-derived immune cells to improve scalability and availability, while ongoing trials continue to address challenges such as graft-versus-host disease, fratricide and limited persistence (Li et al., 2025). At the beginning of last year, a study was published where Induced pluripotent stem-cell (iPSC)-derived chimeric antigen receptor (CAR) natural killer (NK) cell were first tested in humans, during a phase-1 clinical trial for relapsed or refractory B-cell lymphoma, reporting positive outcomes with regards to the safety of the treatment (Ghobadi et al., 2025). Lastly, a novel allogeneic anti-CD70 CAR-T product was tested in a phase-1 trial showing promising results in the treatment of clear cell renal cell carcinoma, adding to the arsenal of cell therapies against solid tumors (Srour et al., 2025).
Source: (Li et al., 2025)
Finally, a special mention goes to Astraveus, which in 2025 has pushed forward CAR-T manufacturing. Astraveus has released a fully automated benchtop microfluidic end-to-end cell factory, enabling rapid (≈26-hour) CAR-T production with transduction efficiency, viability and purity comparable to conventional methods, pointing toward faster and more standardized cell therapy deployment (Peeck & Ghinatti, 2025).
Beyond cancer treatment, cell-based therapies are making enormous steps for many other applications! I’m going to highlight a few more of these below:
Parkinson’s Disease (Stem Cell–Derived Dopaminergic Neurons): A pioneering regenerative approach reached late-stage trials. Bemdaneprocel (BlueRock/Bayer) – implanted dopamine-producing neurons derived from pluripotent stem cells – showed long-term safety and cell engraftment in an initial Phase 1 study, with patients tolerating the implants and imaging suggesting the new neurons survived and integrated into the brain. In 2025, the therapy entered a global Phase 3 trial (exPDite-2) – the first large-scale study of a stem-cell replacement therapy in Parkinson’s. If successful, bemdaneprocel could become the first treatment to restore lost neuronal function in PD rather than just manage symptoms.
Type 1 Diabetes (Islet Cell Replacement – VX-880/Zimislecel): 2025 saw dramatic progress toward a functional cure for T1D using stem cell-derived islet cells. Vertex Pharmaceuticals’ zimislecel (VX-880), pancreatic islet cells derived from stem cells, reported Phase 1/2 trial results at the ADA meeting: all participants showed graft engraftment and C-peptide production, and 10 of 12 patients (83%) achieved insulin independence one year after a single infusion. Patients had HbA1c <7% with no severe hypoglycemia, indicating restored glycemic control (Rickels et al., 2025). While immunosuppression is required (and led to some serious infections), these findings (simultaneously published in NEJM) demonstrate the therapy’s transformative potential. A Phase 3 trial is underway in ~50 patients to confirm efficacy, and Vertex is also exploring gene-edited islet cells that could evade immune rejection in future.
Alzheimer’s Disease (MSC Therapy – Laromestrocel): In regenerative medicine for neurology, an mesenchymal stem cell (MSC) product showed encouraging signs in mild Alzheimer’s disease. Longeveron’s laromestrocel (Lomecel-B), an allogeneic MSC therapy, received an FDA RMAT designation and in a Phase 2 trial (CLEAR MIND) demonstrated reduced neuroinflammation on MRI and slower hippocampal atrophy compared to placebo. Treated patients had preserved hippocampal volume correlating with better clinical outcomes, suggesting the cells’ anti-inflammatory and pro-regenerative effects in the brain. Cognitive function and quality of life improved in initial data, positioning laromestrocel as a novel disease-modifying approach for Alzheimer’s pending larger trials.
Systemic Lupus Erythematosus (Allogeneic CAR T Cells – FT819): A breakthrough therapy applied CAR T-cell technology to autoimmunity. FT819 (Fate Therapeutics) is a first-in-kind CAR T made from an induced pluripotent stem cell line, an off-the-shelf product targeting CD19 B cells, repurposed to wipe out the autoreactive B cells that drive lupus. In 2025, the FDA granted it Regenerative Medicine Advanced Therapy (RMAT) status after early Phase 1 use showed promising results. The first lupus patient treated (at UCI) experienced a striking reduction of lupus symptoms within weeks, the first meaningful remission in nearly two decades of disease. This patient became fully functional with resolution of fevers, joint pain, nephritis, and fatigue. The allogeneic CAR T appears to be tolerated thus far without intensive chemo or hospital isolation. FT819’s early success in lupus (and ongoing trials in myositis, sclerosis, and vasculitis) highlights the potential for CAR T beyond oncology to autoimmune applications.
HIV Cure Research (Personalized T-Cell Therapy – HST-NEET): A Phase 1 trial provided a hopeful step toward an HIV cure using ex vivo expanded T cells. Researchers at Children’s National engineered each patient’s own T cells to recognize conserved HIV epitopes and infused them into 6 people with HIV. The results, published in Nature Communications, shows that therapy was to be safe with no serious adverse effects (Sohai et al., 2025). Early signs of efficacy emerged in 2 patients the treatment boosted HIV-specific T cell and antibody responses, and in another 2 it led to a measurable drop in latent HIV reservoir cells, indicating the hidden virus was being attacked. The infused T cells persisted in circulation for up to 40 weeks, patrolling for HIV. While not yet a cure, these findings show that a targeted cell therapy can reduce the virus lurking in the body and train the immune system to better fight HIV. This personalized T-cell strategy is now moving into expanded trials and combination approaches (e.g., with latency-reversing agents) to further shrink the HIV reservoirs.
Overall, cell therapies are rapidly expanding not only within oncology, but are also making their way through clinical studies for new indications. The field of Cell therapy continues to be exciting with lots of exciting ongoing applications and future potential.
These newsletters take significant effort to put together and are totally for the reader’s benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Abdin, S. M., Paasch, D., & Lachmann, N. (2024). CAR macrophages on a fast track to solid tumor therapy. Nature Immunology, 25(1), Article 1. https://doi.org/10.1038/s41590-023-01696-7
Benthani, F., Upadhaya, S., & Zhou, A. (2025). Cancer cell therapies: Global clinical trial trends and emerging directions. Nature Reviews Drug Discovery, 24(12), 898–899. https://doi.org/10.1038/d41573-025-00180-1
Escobar, G., Berger, T. R., & Maus, M. V. (2025). CAR-T cells in solid tumors: Challenges and breakthroughs. Cell Reports Medicine, 6(11), 102353. https://doi.org/10.1016/j.xcrm.2025.102353
Garvey, W.T., Blüher, M., Osorto Contreras, C.K., Davies, M.J., Winning Lehmann, E., Pietiläinen, K.H., Rubino, D., Sbraccia, P., Wadden, T., Zeuthen, N. and Wilding, J.P., 2025. Coadministered cagrilintide and semaglutide in adults with overweight or obesity. New England Journal of Medicine.
Ghobadi, A., Bachanova, V., Patel, K., Park, J. H., Flinn, I., Riedell, P. A., Bachier, C., Diefenbach, C. S., Wong, C., Bickers, C., Wong, L., Patel, D., Goodridge, J., Denholt, M., Valamehr, B., Elstrom, R. L., & Strati, P. (2025). Induced pluripotent stem-cell-derived CD19-directed chimeric antigen receptor natural killer cells in B-cell lymphoma: A phase 1, first-in-human trial. The Lancet, 405(10473), 127–136. https://doi.org/10.1016/S0140-6736(24)02462-0
Jastreboff, A.M., Aronne, L.J., Ahmad, N.N., Wharton, S., Connery, L., Alves, B., Kiyosue, A., Zhang, S., Liu, B., Bunck, M.C. and Stefanski, A., 2022. Tirzepatide once weekly for the treatment of obesity. New England Journal of Medicine, 387(3), pp.205-216.
Li, Y.-R., Zhu, Y., Fang, Y., Lyu, Z., & Yang, L. (2025). Emerging trends in clinical allogeneic CAR cell therapy. Med, 6(8), 100677. https://doi.org/10.1016/j.medj.2025.100677
Peeck, L. H., & Ghinatti, G. (2025). Small is powerful: Demonstrating the potential of a novel microfluidic benchtop cell factory to optimize and scale CAR-T manufacturing. Cytotherapy, 27(5, Supplement), S195–S196. https://doi.org/10.1016/j.jcyt.2025.03.395
Reiss, K. A., Angelos, M. G., Dees, E. C., Yuan, Y., Ueno, N. T., Pohlmann, P. R., Johnson, M. L., Chao, J., Shestova, O., Serody, J. S., Schmierer, M., Kremp, M., Ball, M., Qureshi, R., Schott, B. H., Sonawane, P., DeLong, S. C., Christiano, M., Swaby, R. F., … Abdou, Y. (2025). CAR-macrophage therapy for HER2-overexpressing advanced solid tumors: A phase 1 trial. Nature Medicine, 31(4), 1171–1182. https://doi.org/10.1038/s41591-025-03495-z
Rickels MR, Witkowski P, Reichman TW, et al. 140-OR - Durable Glycemic Control and Elimination of Exogenous Insulin Use with VX-880 in Patients with Type 1 Diabetes (T1D)—VX-880-101 (FORWARD). Presented at ADA 2025; June 20-23; Chicago, Illinois. Presentation #140-ORRubino D, Abrahamsson N, Davies M, Hesse D, Greenway FL, Jensen C, Lingvay I, Mosenzon O, Rosenstock J, Rubio MA, Rudofsky G. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: the STEP 4 randomized clinical trial. Jama. 2021 Apr 13;325(14):1414-25.
Shokati, A., Sanjari-Pour, M., Akhavan Rahnama, M., Hoseinzadeh, S., Vaezi, M., & Ahmadvand, M. (2025). Allogeneic CART progress: Platforms, current progress and limitations. Frontiers in Immunology, 16. https://doi.org/10.3389/fimmu.2025.1557157
Sohai, D.K., Keller, M.D., Hanley, P.J., Hoq, F., Kukadiya, D., Datar, A., Reynolds, E., Copertino, D.C., Lazarski, C., McCann, C.D. and Tanna, J., 2025. Autologous HIV-specific T cell therapy targeting conserved epitopes is well-tolerated in six adults with HIV: an open-label, single-arm phase 1 study. Nature communications, 16(1), p.4510.
Srour, S. A., Chahoud, J., Drakaki, A., Curti, B. D., Gibney, G. T., Pal, S. K., Tang, L., Charmsaz, S., Atwell, J., Robbins, P. B., Williams, C., Ghatta, S., Severyn, C., Le Gall, J., Tannir, N. M., & Kotecha, R. R. (2025). ALLO-316 in advanced clear cell renal cell carcinoma (ccRCC): Updated results from the phase 1 TRAVERSE study. Journal of Clinical Oncology, 43(16_suppl), 4508–4508. https://doi.org/10.1200/JCO.2025.43.16_suppl.4508
Vitanza, N. A., Ronsley, R., Choe, M., Seidel, K., Huang, W., Rawlings-Rhea, S. D., Beam, M., Steinmetzer, L., Wilson, A. L., Brown, C., Beebe, A., Lindgren, C., Gustafson, J. A., Wein, A., Holtzclaw, S., Hoeppner, C., Goldstein, H. E., Browd, S. R., Hauptman, J. S., … Jensen, M. C. (2025). Intracerebroventricular B7-H3-targeting CAR T cells for diffuse intrinsic pontine glioma: A phase 1 trial. Nature Medicine, 31(3), 861–868. https://doi.org/10.1038/s41591-024-03451-3
West, S., Scragg, J., Aveyard, P., Oke, J.L., Willis, L., Haffner, S.J., Knight, H., Wang, D., Morrow, S., Heath, L. and Jebb, S.A., 2026. Weight regain after cessation of medication for weight management: systematic review and meta-analysis. bmj, 392.
Wharton S, Lingvay I, Bogdanski P, Duque do Vale R, Jacob S, Karlsson T, Shaji C, Rubino D, Garvey WT. Oral semaglutide at a dose of 25 mg in adults with overweight or obesity. New England Journal of Medicine. 2025 Sep 18;393(11):1077-87.











Very cool. And thorough…thank you!
Thank you! From @Neuromics.