Rewiring the Brain: Phase I Data Promising for Parkinson's Cell-Based Therapy
The Science and Skepticism Behind the Latest Parkinson's Trial
Introduction
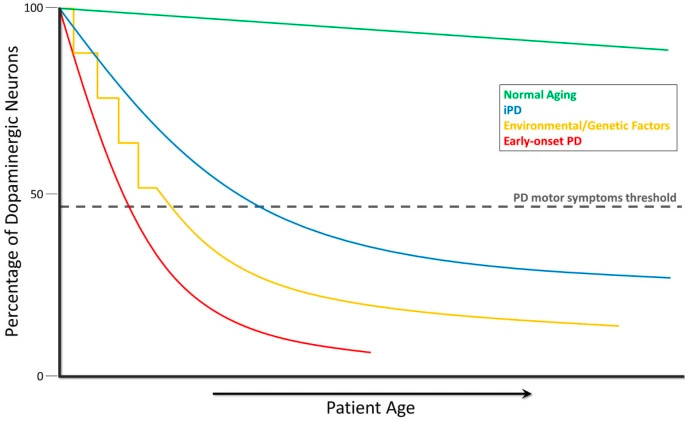
Parkinson's disease is a neurodegenerative disorder affecting over 10 million people globally. The disease progressively damages dopaminergic neurons in the brain, which are key nerve cells responsible for the production of dopamine. This leads to a systemic decrease in dopamine levels, a neurotransmitter crucial for regulating mood and movement (Figure 1). As the destruction of dopaminergic neurons reaches a critical threshold, symptoms of Parkinson’s disease—such as tremors, muscle rigidity, and slowness of movement—begin to manifest (Figure 2).
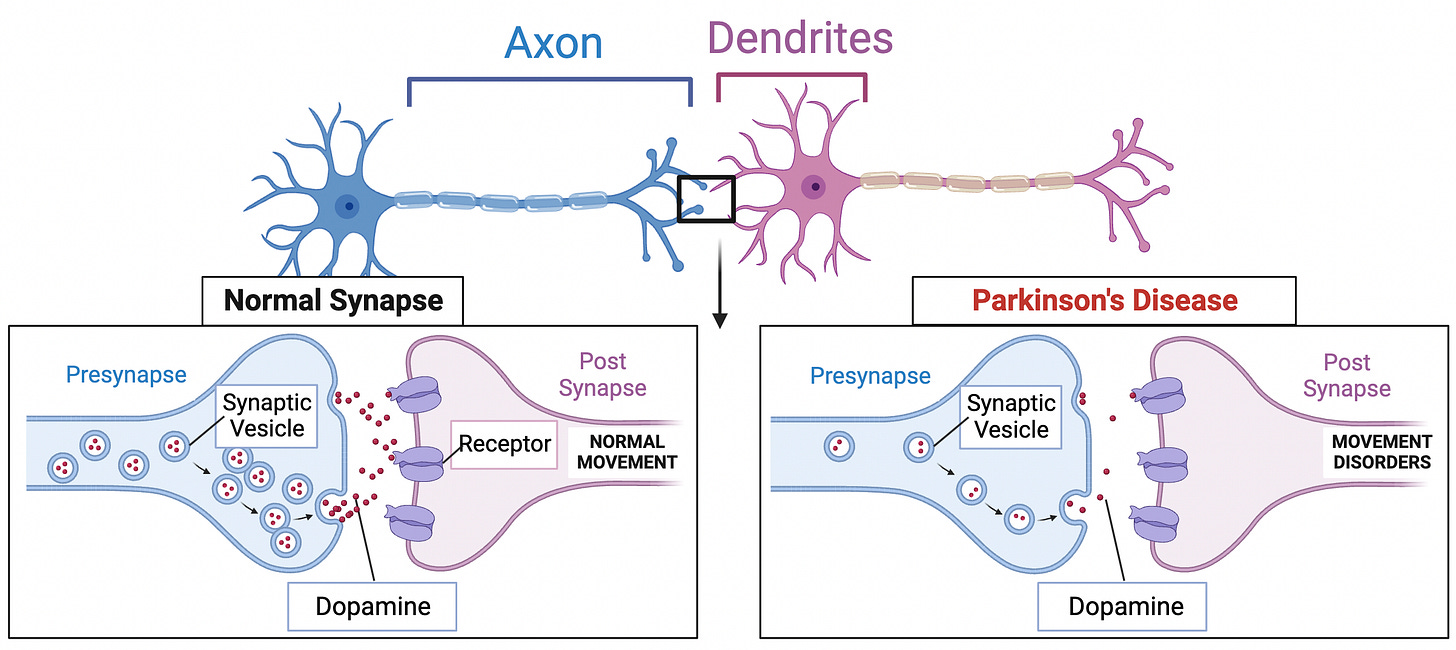
Researchers have been making strides toward replacing these lost dopaminergic neurons. In various animal models of Parkinson’s disease, proof-of-concept work has been performed. One landmark study transplanted dopaminergic neurons into the brains of monkeys with simulated Parkinson’s disease and demonstrated a reduction in symptoms (Kikuchi, 2017). Recently, BlueRock Therapeutics, a subsidiary of Bayer, claims to have made significant progress in treating this disease by implanting lab-made dopamine-producing neurons into the human brain. While these initial results are promising, they also invite several questions and concerns that merit a balanced and critical discussion.
The Claim
BlueRock Therapeutics, a subsidiary of Bayer, has developed a novel therapeutic known as Bemdaneprocel (BRT-DA01) aimed at treating Parkinson's Disease. This therapeutic is particularly groundbreaking because it consists of dopamine-producing neurons that are derived from embryonic stem cells.
The company recently concluded a small-scale Phase I clinical trial involving 12 patients diagnosed with Parkinson's disease. In clinical research, the primary objective of a Phase I trial is usually to assess the safety and tolerability of a treatment and or treatment. Secondary objectives often focus on gathering preliminary evidence for efficacy, which, in this context, would mean the survival of the implanted neurons and any observed improvement in the symptoms of Parkinson's disease.
As of late June, the clinical trial was completed, and the detailed results were shared at the International Congress on Parkinson’s Disease and Movement Disorder in Copenhagen. According to BlueRock Therapeutics, the trial met its primary objective with resounding success. BRT-DA01 was well-tolerated and presented no major safety issues in all 12 patients through the one-year follow-up period (Regaldo, 2023). Further, while the data on these secondary objectives have not been fully disclosed, the initial reports suggest promising outcomes such as brain scans indicating an increase in dopamine cells and a decrease in "off time," the hours per day when patients felt incapacitated by their symptoms, particularly in the higher dosing treatment group.
Impact (Why You May Care)
The successful primary outcome of BlueRock Therapeutics' Phase I clinical trial represents a significant leap forward for cell-based therapy in the field of neurodegenerative disease. If BRT-DA01 continues to show tolerability and efficacy in subsequent, larger trials, it would fundamentally change the way we approach the treatment of Parkinson's Disease. However, the therapy's potential extends well beyond merely managing symptoms to possibly reversing some of the disease's effects by replenishing dopamine levels in the brain.
Moreover, the implications of this research could ripple across various medical disciplines. The technology behind BRT-DA01—using embryonic stem cells to create specialized neurons—could be adapted for other conditions involving neurotransmitter imbalances or neuronal loss. This could include applications for ‘anti-aging’ or ‘longevity’ and diseases like Alzheimer's, Huntington's, and even certain types of depression. In essence, the trial opens the door to a new era of regenerative medicine, where damaged or lost cells could be replaced by lab-grown counterparts. Furthermore, any treatment that improves function in a disease state has the potential to enhance a normal functioning brain.
The success of this trial also adds momentum to the broader field of stem cell research. If embryonic stem cells can be safely and effectively used to treat a condition as complex as Parkinson's, it could validate the utility of stem cell therapies for a range of other medical conditions, from spinal cord injuries to heart diseases.
Is This Real?
While the initial results from BlueRock Therapeutics' Phase I clinical trial are undeniably promising, it's crucial to approach them with a healthy dose of skepticism and tempered expectations. Here are some points to consider:
Sample Size and Duration. The trial involved only 12 patients and lasted for one year. This is not abnormal for such a novel and expensive treatment. However, this size certainly reduces the power of claims regarding safety and impact.
Previous Catastrophes. It's important to note that earlier attempts at cell-based therapies for Parkinson's Disease have not always been successful. Early attempts for cell-based therapies utilized aborted fetal tissue grafts, where, in some cases, patients developed dyskinesias—uncontrollable, involuntary movements—as a side effect of the treatment. It is believed that the variability in these results was due to tissue harvesting and preparation from aborted fetuses (which seems to have its own ethical implications to put it mildly). These past setbacks underscore the need for rigorous, long-term studies before any new therapy can be deemed safe and effective (Fitzpatrick, 2009).
Ethical and Legal Concerns. The use of embryonic stem cells raises ethical questions that could limit the therapy's widespread adoption. In some countries, the derivation of cells from embryos is illegal, which could impede global accessibility to the treatment. However, as with the Primate study referred to in the introduction, iPSCs may present an alternative to embryonic stem cells. This could improve both the reproducibility and remove the ethical considerations.
Lack of detailed data on secondary objectives. While the initial reports suggest promising outcomes for Parkinson’s Disease symptoms, the data on these secondary objectives have not been fully disclosed yet. It is normal for a study to play their cards close to their chest, wanting to make sure results are fully contextualized before sharing. However, we will not truly know the efficacy until this data is shared and Phase II studies commence. In addition, an expert like Roger Barker from the University of Cambridge expressed some caution. Barker called the evidence that the transplanted cells had survived “a bit disappointing,” highlighting the need for more robust data.
Financial Interests. BlueRock Therapeutics is a subsidiary of Bayer, a multinational pharmaceutical company. The financial stakes involved could potentially influence the presentation and interpretation of trial results
Conclusions and Next Steps
The Phase I clinical trial marks a promising but preliminary step in the development of an effective cell-based therapy for Parkinson's Disease. The trial's primary objective of assessing the safety of BRT-DA01 was met, but several questions and concerns remain, including the therapy's long-term efficacy, ethical considerations, and the need for more robust data.
What to Watch For. BlueRock Therapeutics has announced plans for Phase II trials in H1 2024. These will be crucial for further validating the safety and efficacy of BRT-DA01.
Expert Opinions. Keep an eye on reactions from the scientific community, especially from experts like Roger Barker, who have expressed caution about the initial results.
Ethical and Legal Developments. As research progresses, it will be interesting to see how the scientific community and lawmakers address the ethical concerns surrounding the use of embryonic stem cells.
Final Thoughts
I am excited about these results, but they should be viewed with cautious optimism. The field of cell-based therapies for Parkinson's Disease (or any others utilizing embryonic stem cells) is complex and fraught with both ethical and scientific challenges. The work being done by BlueRock Therapeutics is very important and could potentially pave the way for groundbreaking treatments. This is not just for Parkinson's but for a range of neurodegenerative diseases and human longevity.
References
BlueRock's Neuronal Stem Cell Therapy for Parkinson's Disease Is First to Show Positive Results in Phase I Clinical Study - https://www.bluerocktx.com/bluerocks-neuronal-stem-cell-therapy-for-parkinsons-disease-is-first-to-show-positive-results-in-phase-i-clinical-study/
Biotech company implants dopamine-making cells into people's brains https://www.technologyreview.com/2023/08/31/1078728/biotech-dopamine-making-cells-into-peoples-brains/?utm_source=Twitter&utm_campaign=site_visitor.unpaid.engagement&utm_medium=tr_social
Bayer says Parkinson's stem cell therapy improves symptoms in initial trial https://www.reuters.com/business/healthcare-pharmaceuticals/bayer-says-parkinsons-stem-cell-therapy-improves-symptoms-initial-trial-2023-08-28/#:~:text=Bayer%20says%20Parkinson's%20stem%20cell%20therapy%20improves%20symptoms%20in%20initial%20trial,-By%20Ludwig%20Burger&text=FRANKFURT%2C%20Aug%2028%20(Reuters),an%20early%2012%2Dpatient%20trial.
Fitzpatrick, K.M., Raschke, J. and Emborg, M.E., 2009. Cell-based therapies for Parkinson's disease: past, present, and future. Antioxidants & redox signaling, 11(9), pp.2189-2208.
Haas, B.R., Stewart, T.H. and Zhang, J., 2012. Premotor biomarkers for Parkinson's disease-a promising direction of research. Translational neurodegeneration, 1, pp.1-11.
Kikuchi, T., Morizane, A., Doi, D., Magotani, H., Onoe, H., Hayashi, T., Mizuma, H., Takara, S., Takahashi, R., Inoue, H. and Morita, S., 2017. Human iPS cell-derived dopaminergic neurons function in a primate Parkinson’s disease model. Nature, 548(7669), pp.592-596.



Neuro-degeneration is such a complex and complicated illness. However, if this research bodes positive results, the future looks bright!