One Blood Test for 6 Types of Dementia
An AI model uses plasma proteins to diagnose and differentiate forms of dementia.
You’re home visiting the family for Thanksgiving. You notice your father-in-law seems to have some odd memory lapses. You quietly bring it up with other family members who independently noticed similar things. You gently suggest to your mother-in-law that it may be time to consult a neurologist. After spending a couple of months scheduling appointments and sitting on a wait list, the tests were inconclusive.
This speed runs the frustrating medical experience for many aging Americans. Misdiagnosis rates for neurodegenerative diseases range from 25 to 30 percent in specialized dementia clinics (Beach et al., 2012). The problem compounds with age: by 80, roughly 70 percent of patients harbor multiple neurodegenerative pathologies simultaneously (Kapasi et al., 2017). Alzheimer’s tangled with vascular disease. Parkinson’s layered over frontotemporal degeneration. The brain doesn’t pick one disease and commit.
Blood-based tests for Alzheimer’s (p-tau217, amyloid ratios) have made progress, but even in this case, it’s only part of the story. For Parkinson’s, ALS, and frontotemporal dementia, clinical diagnosis still relies on symptoms and exclusion. A confirmatory test with specific biomarkers is either invasive (cerebrospinal fluid), expensive (PET imaging), or unavailable entirely. That gap is where misdiagnosis lives.
If you enjoy these posts, consider subscribing and becoming a part of our growing community!
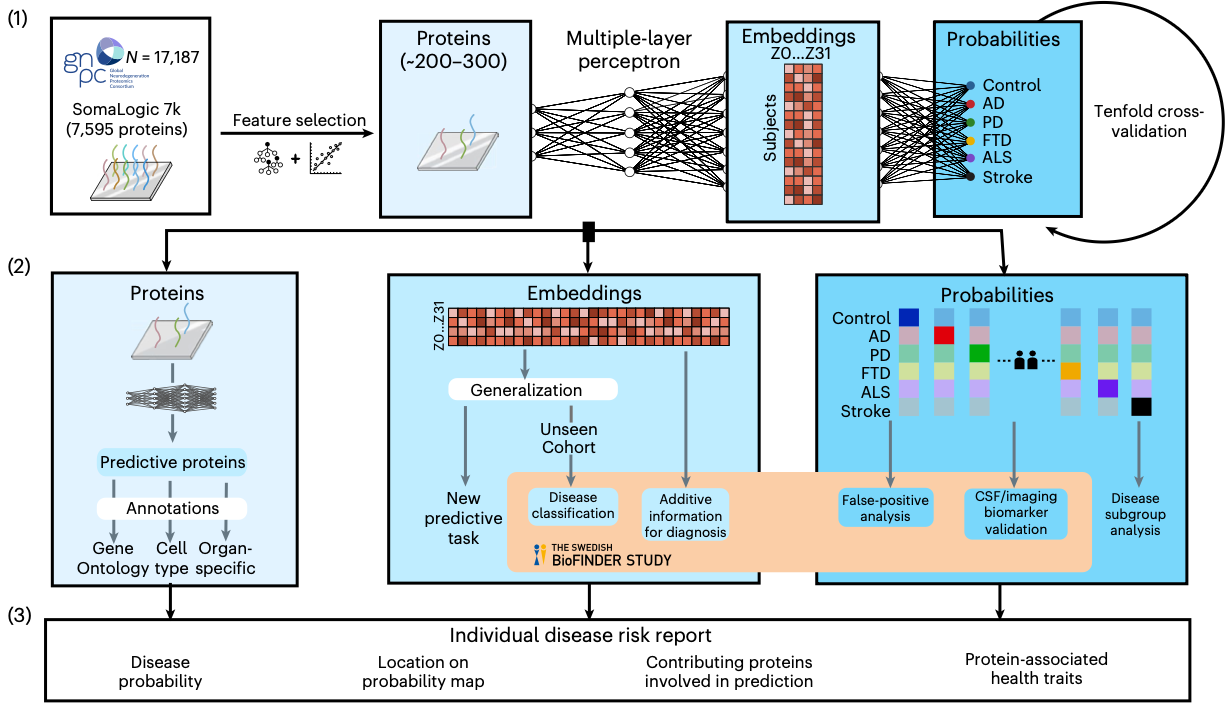
A team at Lund University just made that gap tractable. Published in Nature Medicine, the team trained a deep learning model called ProtAIDe-Dx on plasma proteomics data from over 17,000 patients across 19 clinical sites (An et al., Nature Medicine, 2026). The model reads roughly 7,600 proteins from a single blood draw, then simultaneously generates probabilistic diagnoses for six conditions: Alzheimer’s disease, Parkinson’s disease, frontotemporal dementia, ALS, stroke/TIA, and cognitive health.
ProtAIDe-Dx achieved balanced classification accuracy of 95% for ALS, 92% for Parkinson’s, 83% for cognitively unimpaired controls, 81% for Alzheimer’s, 72% for frontotemporal dementia, and 70% for stroke. For a single blood test with no clinical inputs, those numbers match or exceed the diagnostic accuracy of a general neurologist’s clinical assessment.
The model learns all six conditions simultaneously. Shared biological signals between diseases inform each prediction, and it generates probability scores for every condition in every patient. That means it can flag co-pathology: a patient scoring high for both Alzheimer’s and Parkinson’s likely has both.
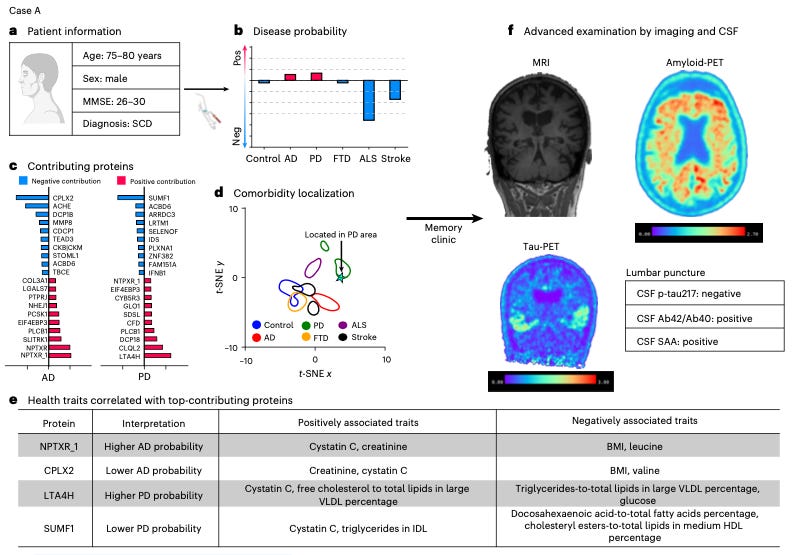
The strongest clinical result came from tracking patients over time. ProtAIDe-Dx predictions stratified future cognitive decline better than clinical diagnosis alone. Patients the model flagged as likely Alzheimer’s declined faster on cognitive testing regardless of their clinical label at baseline. Some apparent false positives may have been catching preclinical pathology that the clinical label missed.
The team validated this externally in BioFINDER-2, a memory clinic cohort of over 1,700 patients with biomarker-confirmed diagnoses. Patients flagged for elevated Alzheimer’s probability were more likely to test positive for amyloid and tau on confirmatory imaging. Elevated stroke probability tracked with white matter lesion burden on MRI. When combined with standard clinical biomarkers, the model significantly improved differential diagnosis accuracy, especially for non-Alzheimer’s dementias where existing blood tests fall short.
Beyond diagnosis, the model identified a set of proteins that discriminate healthy aging from neurodegeneration across all six conditions. One of them detoxifies a byproduct of glucose metabolism that drives protein damage and accumulates with age. Its appearance suggests that metabolic vulnerability may be a shared upstream signal across neurodegenerative diseases, one where the failure to clear cellular damage accumulates into vulnerability.
Healthcare systems require disease diagnosis before administering therapeutics, especially when the care is expensive. For Alzheimer’s disease, lecanemab and donanemab require biomarker-confirmed amyloid status before treatment can begin. That confirmation currently means several months of navigating patient visits to reach a PET scan, averaging $3,000, or a lumbar puncture. A SomaScan proteomics panel runs roughly $950 per sample and returns six probabilistic diagnoses. The economics of triage favor blood. A test that flags which patients need expensive confirmatory imaging, which should enter a clinical trial, and which carry preclinical pathology worth monitoring could reshape how neurology clinics process their intake queues. The family that spent two months on a wait list for an inconclusive answer could have six probabilities from a single tube of blood for a third of the cost and none of the headache.
These newsletters take significant effort to put together and are totally for the reader’s benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References
Beach TG, Monsell SE, Phillips LE, Kukull W. Accuracy of the clinical diagnosis of Alzheimer disease at National Institute on Aging Alzheimer Disease Centers, 2005-2010. J Neuropathol Exp Neurol. 2012;71:266-273. doi: 10.1097/NEN.0b013e31824b211b
Kapasi A, DeCarli C, Schneider JA. Impact of multiple pathologies on the threshold for clinically overt dementia. Acta Neuropathol. 2017;134:171-186. doi: 10.1007/s00401-017-1717-7
An L, Pichet Binette A, Hristovska I, et al. A deep joint-learning proteomics model for diagnosis of six conditions associated with dementia. Nature Medicine. 2026. doi: 10.1038/s41591-026-04303-y