IVF's Next Frontier: Harnessing iPSC-Derived Oocytes in Reproductive Medicine
Developing human egg cells from skin biopsies is on the horizon.
Foreword:
In a previous article, we took a deep dive into in vitro fertilization (IVF) advancements and how these impacted the probability of a resulting birth. In the scope of that article, the technologies we discussed primarily centered around the selection of the healthiest embryos for implantation. However, there are many other interesting emerging technologies that could greatly enhance the field of reproductive medicine. One such technology I would like to discuss in greater depth today is the ability to synthetically generate oocytes (egg cells) from induced pluripotent stem cells (iPSCs). For those who are unfamiliar with iPSCs, this may sound like a bit like a word salad. But don’t worry, this is still new technology. By the end of this article, I will help you to be literate in iPSC technology and hopefully convey the implications of this enormous breakthrough for IVF.
Introduction
Infertility affects a significant portion of the global population, posing emotional, physical, and sometimes social challenges to those who experience it. In a previous article, we reviewed how IVF has emerged as a powerful tool for treating infertility. Furthermore, we discussed the overall process of IVF, which is summarized in Figure 1. This begins with the stimulation and retrieval of egg cells from a prospective woman and ends with their fertilization and postimplantation for embryo development. Unbeknownst to me at the time of writing the original article, the procedure of egg retrieval can be quite invasive and even painful (Nakagawa et al., 2015)! Furthermore, the harvested oocytes are typically the rate-limiting material for IVF procedures, where only between 7-15 oocytes are harvested per treatment cycle. However, this bottleneck could become a thing of the past thanks to iPSCs.
Induced pluripotent stem cells (iPSCs) represent a pivotal breakthrough in the field of regenerative medicine. iPSCs are derived from adult cells (often taken from a skin biopsy) and reprogrammed to an embryonic stem cell-like state, offering a potentially unlimited source of any cell type for therapeutic purposes. While these iPSCs have an obvious therapeutic potential in addressing the issue of oocyte availability, quantity, and quality, there is a catch: these iPSCs must be differentiated from their stem cell state to the terminal cell type. But before we discuss this further, let’s talk a bit more in-depth about iPSCs.
Understanding the basics of iPSCs
Induced pluripotent stem cells (iPSCs) are a marvel of modern science, offering a new realm of possibilities in medical research. They were first successfully developed in 2006 by Shinya Yamanaka and his team, who discovered that adult somatic cells could be reprogrammed into embryonic stem cell-like states. This reprogramming is achieved by introducing a set of specific transcription factors, often referred to as Yamanaka factors, into the cells. These factors (typically Oct3/4, Sox2, c-Myc, and Klf4) effectively reset the mature cells into a pluripotent state, allowing them to differentiate into virtually any cell type.
The significance of iPSCs in medical research is profound. Unlike embryonic stem cells, which require the destruction of an embryo, iPSCs can be derived from adult cells, such as skin or blood cells. This addresses major ethical concerns associated with stem cell research and has revolutionized the field by providing a more acceptable alternative. iPSCs are not only crucial for understanding disease mechanisms and drug testing but also hold immense potential for regenerative medicine, tissue engineering, and personalized therapies. The ability to create patient-specific iPSC lines allows for the study of diseases and the development of treatments in a way that is tailored to individual genetic profiles, reducing the risk of immune rejection and enhancing treatment efficacy.
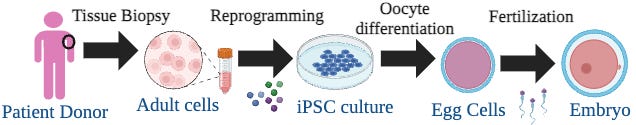
Okay, so how do we go from an iPSC to an oocyte (Figure 2)?
The path to iPSC-Derived Oocytes
One of the most remarkable applications of iPSC technology has been in the field of reproductive biology, particularly in generating oocytes from iPSCs. The majority of breakthroughs have come from studies conducted on mice. In these studies, researchers first demonstrated that iPSCs could be differentiated into primordial germ cell-like cells (PGCLCs), which are the precursors to oocytes and sperm.
Building on this, a key study by Hayashi and colleagues in 2012 successfully produced functional oocytes from mouse iPSCs. These iPSC-derived oocytes were then fertilized with sperm and implanted into female mice for gestation. Incredibly, these embryos matured to produce healthy, fertile offspring, demonstrating the functional viability of the starting oocytes (Figure 3). This was obviously an enormous milestone for stem cell biology and opened up new avenues in reproductive medicine.
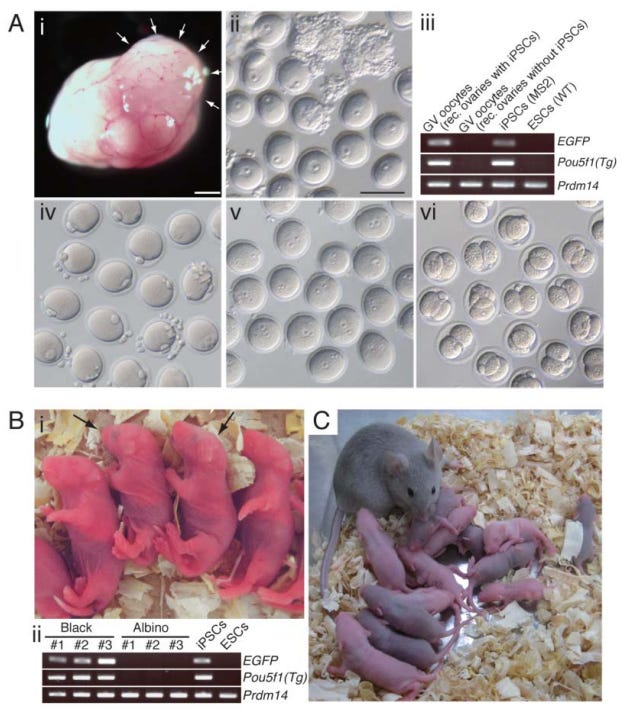
Okay, but is any of this possible in humans?
Converting human iPSCs to oocytes has proven to be more challenging. However, significant progress has still been made. Studies have successfully differentiated human iPSCs into PGCLCs, the precursors to oocytes (Yang et al, 2022). However, the oocyte-like cells generated from these did not express certain key proteins, indicating they could not substitute for fully functional oocytes in vivo. This suggests additional work is still required for the proper maturation of functional oocytes. One additional study recently showed the successful induction of meiotic oocytes from the embryonic stem cells of cynomolgus monkeys (Gyobu-Motani et al., 2023), which share many physiological traits with humans. This represents a significant step forward in understanding stem cell differentiation into oocytes in humans. Overall, there have been promising developments in the differentiation of iPSCs into oocyte-like cells and PGCLCs, but the generation of fully functional human oocytes from iPSCs is likely still a bit into the future.
iPSC-Derived Oocytes: Potential Impact on IVF
The ability to generate oocytes from induced pluripotent stem cells (iPSCs) has the potential to revolutionize the field of in vitro fertilization (IVF). Traditionally, IVF has been limited by the number and quality of oocytes available for retrieval from patients. With the advent of iPSC technology, these limitations could be significantly mitigated, opening up new possibilities for individuals and couples facing infertility.
Expanding the Pool of Available Oocytes - One of the most immediate impacts of iPSC-derived oocytes would be the expansion of the available pool of oocytes for IVF procedures. This would be particularly beneficial for patients with low ovarian reserve or those who produce a limited number of eggs. By creating oocytes from the patient's own cells, it would be possible to generate a larger number of viable eggs, increasing the chances of successful fertilization and pregnancy.
Enhancing Oocyte Quality - Another advantage of iPSC-derived oocytes is the potential for enhanced quality. As oocytes age, their quality diminishes, which can lead to lower success rates in IVF. iPSC technology offers a way to create oocytes that are genetically younger, potentially improving the quality of embryos and increasing the success rates (live births) following IVF treatments.
Male-derived oocytes may enable offspring for same-sex couples - If oocytes can be generated from female iPSCs, might they be generated from male cells as well? This was explored by Murakami et al, who found the answer to be ‘yes’. In their study (Murakami et al, 2023), they successfully generated functional oocytes from male mouse cells by converting XY chromosome set to XX without an additional Y chromosome in pluripotent stem cells. This chromosomal alteration eliminated trisomy 16 and allowed for the differentiation of euploid XX pluripotent stem cells into mature oocytes in culture. Incredibly, the conversion of induced pluripotent stem cells from sexually mature male mice into fully potent oocytes eventually led to successful offspring after fertilization.
Ethical and Social Implications - The development of iPSC-derived oocytes also raises important ethical and social considerations. While this technology offers hope to many struggling with infertility, it also prompts questions about the ethics of creating gametes in the lab, the potential for genetic manipulation, and the implications for identity and parenthood. This doesn’t even touch on topics such as ‘where does life begin’. These and other such existential questions will inevitably arise along with the general ethics of fertilization of more embryos than one plans to use for implantation. In a recent ruling from the Alabama supreme court, personhood has been extended to fertilized embryos in IVF clinics. The goal here is not to debate the politics of this but to inform the overall conversation.
Challenges and Next Steps
While the potential of iPSC-derived oocytes is immense, there are significant challenges that must be overcome before this technology can be applied in clinical IVF practices.
Differentiation Efficiency: Current methods for differentiating iPSCs into oocytes are inefficient and unreliable. Scientists must refine these protocols to consistently produce oocytes that are indistinguishable from their natural counterparts in terms of functionality and genetic stability.
Safety and Quality Control: Any oocytes generated from iPSCs must undergo rigorous testing to ensure they do not carry harmful mutations or epigenetic modifications that could impact the health of the baby. This includes developing new assays for assessing the genetic and epigenetic integrity of iPSC-derived oocytes.
Regulatory Hurdles: Before iPSC-derived oocytes can be used in IVF treatments, they must clear extensive regulatory scrutiny to ensure their safety and efficacy. Establishing a regulatory framework that can adapt to the rapid advancements in iPSC technology will be crucial.
Conclusion
The development of functional iPSC-derived oocytes represents a monumental breakthrough in the realm of reproductive medicine, offering a novel solution for individuals facing fertility challenges. Although this methodology has already shown promise in mouse models, further advancements are necessary to translate these findings into human applications. Despite the significant obstacles that lie ahead, the field stands on the brink of a major breakthrough. As we move forward, it will be crucial to address the technical, safety, and ethical considerations associated with iPSC-derived oocytes, ensuring that the promise of this groundbreaking technology can be fully realized in a manner that respects the dignity and rights of all humans.
References:
https://publichealth.jhu.edu/2024/the-alabama-supreme-courts-ruling-on-frozen-embryos (Accessed 03MAR24).
Murakami, K., Hamazaki, N., Hamada, N., Nagamatsu, G., Okamoto, I., Ohta, H., Nosaka, Y., Ishikura, Y., Kitajima, T.S., Semba, Y. and Kunisaki, Y., 2023. Generation of functional oocytes from male mice in vitro. Nature, 615(7954), pp.900-906.
Aizawa, E., Ozonov, E.A., Kawamura, Y.K., Dumeau, C.E., Nagaoka, S., Kitajima, T.S., Saitou, M., Peters, A.H. and Wutz, A., 2023. Epigenetic regulation limits competence of pluripotent stem cell‐derived oocytes. The EMBO Journal, p.e113955.
Gyobu‐Motani, S., Yabuta, Y., Mizuta, K., Katou, Y., Okamoto, I., Kawasaki, M., Kitamura, A., Tsukiyama, T., Iwatani, C., Tsuchiya, H. and Tsujimura, T., 2023. Induction of fetal meiotic oocytes from embryonic stem cells in cynomolgus monkeys. The EMBO Journal, 42(9), p.e112962.
Hayashi, K., Ohta, H., Kurimoto, K., Aramaki, S. and Saitou, M., 2011. Reconstitution of the mouse germ cell specification pathway in culture by pluripotent stem cells. Cell, 146(4), pp.519-532.
Hayashi, K., Ogushi, S., Kurimoto, K., Shimamoto, S., Ohta, H. and Saitou, M., 2012. Offspring from oocytes derived from in vitro primordial germ cell–like cells in mice. Science, 338(6109), pp.971-975.
Nakagawa, K., Nishi, Y., Kaneyama, M., Sugiyama, R., Motoyama, H. and Sugiyama, R., 2015. The effect of a newly designed needle on the pain and bleeding of patients during oocyte retrieval of a single follicle. Journal of Reproduction & Infertility, 16(4), p.207.
Yang, S., Liu, Z., Wu, S., Zou, L., Cao, Y., Xu, H., Huang, J., Tian, Q., Wu, F., Li, P. and Peng, S., 2022. Meiosis resumption in human primordial germ cells from induced pluripotent stem cells by in vitro activation and reconstruction of ovarian nests. Stem Cell Research & Therapy, 13(1), pp.1-11.



That's fascinating. It doesn't set off any ethical alarm bells, either. Maybe it should; maybe a case could be made that instead of putting all that energy, time, and expense into making babies, people who want kids should be encouraged adopt them. That case won't be made by me, though. Could unwanted genetic tinkering happen? Can't it happen now, with current IVF procedures? It seems as though if anything the ethical problems cited with IVF technology should be mitigated with iPSC oocytes. The whole "every sperm is sacred" problem goes away. Although not the fertilized embryo problem, I guess.
Speaking of sperm, is anyone trying to make male gametes, or is there an impregnable (heh) technical barrier to that?
Keep up the good articles!