Inverse Vaccines: A Revolutionary Approach to Treating Autoimmune Diseases
The Promise and Challenges of Tolerogenic Therapies

Foreword:
My mother, in her early 20s, began developing a variety of medical symptoms that didn’t seem normal to her. She would often feel extremely fatigued, have strange swelling in her joints, and have severe reactions to small changes in temperature (Raynaud’s phenomenon). Some time later she would also develop a severe rash on her face. She went to several doctors who were baffled; one doctor suggested that she may have an ‘ear infection.’ After several such visits, a doctor noticed some abnormalities in her blood work. Eventually, she was referred to a rheumatologist who diagnosed her with systemic lupus erythematosus, commonly referred to as ‘Lupus.’ In a related story, my cousin and I both started losing pigment in our hands, feet, and face during our late teens and early twenties. In both our cases, the diagnosis was vitiligo.
What do these cases have in common?
These seemingly unrelated diseases are all classic cases of autoimmune disorders. These are immune disorders where the body’s immune system attacks its own tissues. The primary differences in these diseases are the body regions that the immune system mistakenly targets (or loses tolerance for). The treatments for this family of diseases had been nearly nonexistent; my cousin and I were advised to modify our diets and manage our stress levels. In more severe cases like my mother's, it is common to be prescribed powerful nonspecific immunosuppressant drugs that have a variety of horrific side effects. However, it appears that medical research may be on the cusp of a significant breakthrough: the modulation of the immune system through 'inverse vaccines', also known as ‘tolerogenic vaccines’. Unlike traditional vaccines that boost our immune response against invaders, 'inverse vaccines' aim to dial it back, teaching our immune system to stop attacking our own body. This post aims to introduce inverse vaccines, assess their claims, discuss the potential impact, and look for where we should be skeptical.
Introduction
Autoimmune diseases have long been a complex puzzle for medical researchers and a source of daily challenges for patients. In these conditions, the immune system mistakenly attacks the body's own cells, leading to a range of symptoms, from mild to severe. For instance, in vitiligo, melanocytes (the pigment-producing cells in the skin) are attacked and destroyed, resulting in the gradual loss of pigmentation (Michael Jackson famously had vitiligo). In the case of lupus, this results in chronic inflammation, which can localize in certain tissues. There are many such autoimmune disorders. While my experience with vitiligo is primarily aesthetic, others face life-altering symptoms. Multiple sclerosis, for example, attacks the protective covering of nerve fibers, leading to neurological issues.
For decades, treatments for autoimmune diseases have remained largely unchanged. The traditional treatments for severe autoimmune diseases often involve potent systemic immunosuppressive drugs like corticosteroids, which have severe side effects. However, we may be on the verge of a breakthrough. What if we could reprogram the immune system instead of simply suppressing it? A recent study from the University of Chicago's Pritzker School of Molecular Engineering offers a groundbreaking approach: an "inverse vaccine" designed to treat multiple sclerosis and potentially other autoimmune diseases (Williams, 2023 and Tremain, 2023).
Traditional vaccines work by introducing a weakened or inactivated form of a pathogen, or a piece of it, into the body (Figure 1). The presentation of this foreign object (antigen) trains the immune system to recognize and fight the pathogen. One mechanism of this is recognition through antigen-presenting cells, which take up the antigen, process it, and present fragments of it on their surface. T cells then recognize these antigen fragments and become activated, proliferating and differentiating into other types of t cells. In contrast to this, an 'inverse vaccine' aims to do the opposite: it seeks to train the immune system to stop recognizing specific antigens "reprogramming" it to be more tolerant. This is a revolutionary concept that flips the conventional understanding of vaccines on its head.
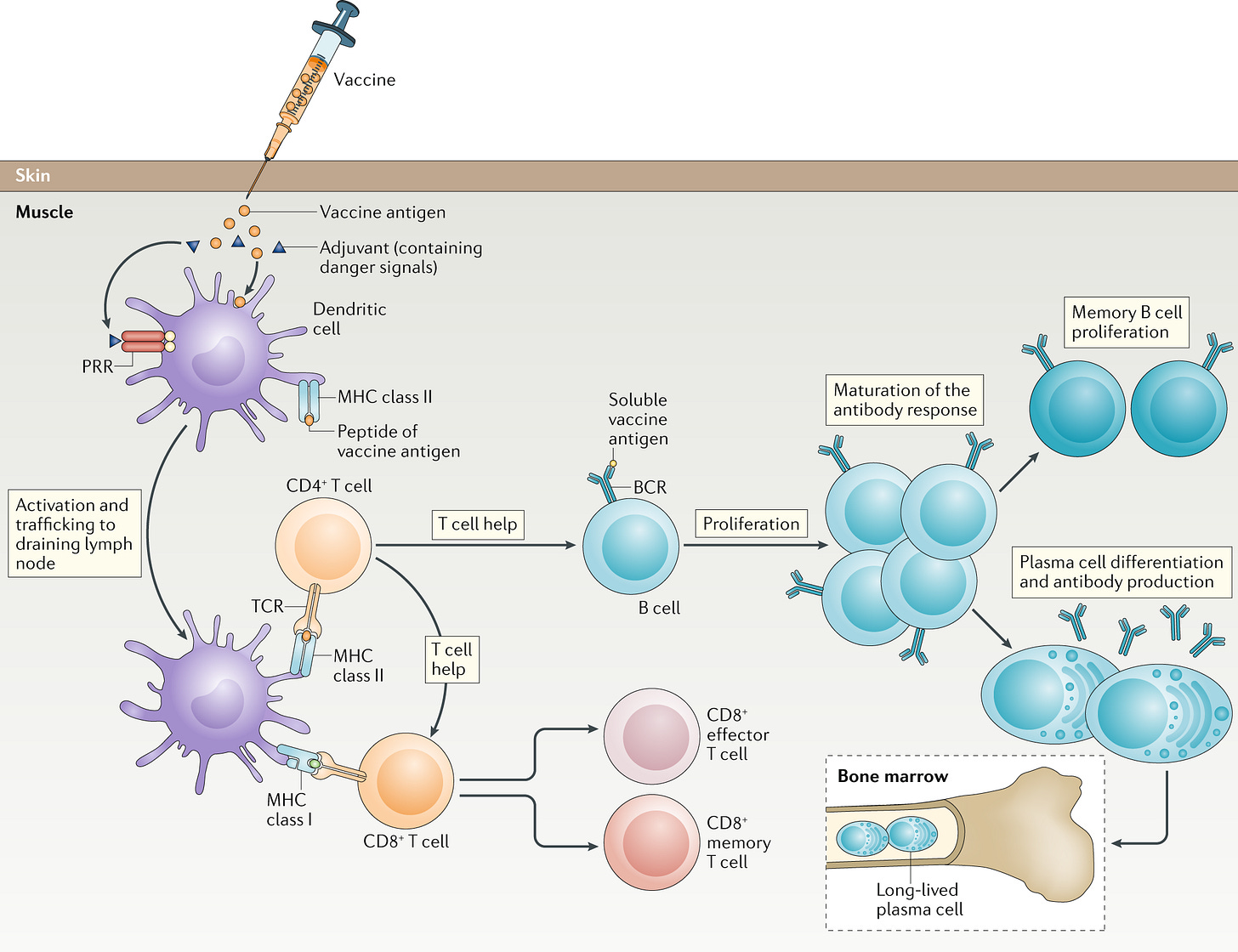
The idea behind inverse vaccines is to use specialized molecules that can attach to specific antigens—proteins that trigger an immune response. By doing so, these molecules can modulate the behavior of T cells, which are often the culprits in autoimmune diseases. Instead of attacking the body's own tissues, the T cells learn to recognize them as 'self' and leave them alone. This approach has the potential to provide a more targeted and less harmful alternative to current immunosuppressive therapies, setting the stage for a new era in the treatment of autoimmune diseases.
Claim(s)
The study presents a groundbreaking approach to treating autoimmune diseases like multiple sclerosis by reprogramming the immune system. Researchers used a specialized molecule called pGal, which they attached to an antigen. This pGal–antigen combination was administered intravenously to mice and non-human primates. The results were striking: the treatment significantly reduced the number of reactive T cells, which are key players in autoimmune diseases.
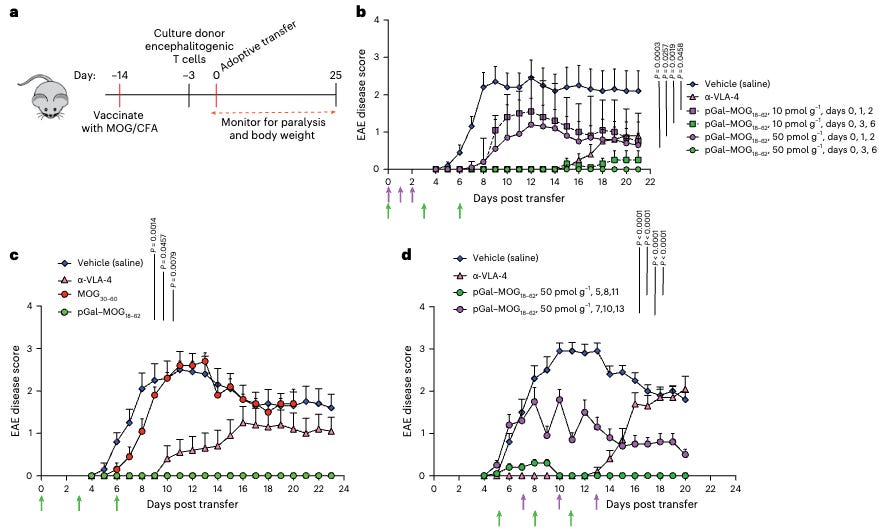
But it didn't stop there. The treatment also changed the internal "software" of these T cells, making them less likely to cause inflammation. This was confirmed through RNA sequencing, which showed that genes associated with inflammation were downregulated, while those linked to immune tolerance were upregulated. Importantly, this approach was effective in both "memory" and "effector" stages of the immune response, meaning it could potentially be used in ongoing autoimmune reactions and not just as a preventive measure. The treatment was tested in mouse models mimicking multiple sclerosis (Figure 2) and even showed promise in suppressing vaccine responses in non-human primates.
How Does the Inverse Vaccine Work?
Natural Liver Function: The liver plays a crucial role in preventing autoimmune reactions. When cells in our body die naturally, the liver marks the molecules from these broken-down cells with "do not attack" flags. This is the liver's way of telling the immune system, "These are our own cells, don't attack them."
Inverse Vaccine Mechanism: The researchers took advantage of this natural liver function. They combined an antigen (the molecule that the immune system wrongly attacks in autoimmune diseases) with a molecule that resembles a fragment of an aged cell. This fragment is something the liver would recognize as friendly.
Result: When this combined molecule (antigen + aged cell fragment) is introduced into the body, the liver marks it with the "do not attack" flag. This means that the immune system is now trained to see the antigen as friendly and stops attacking it.
Practical Application: The researchers tested this approach in a disease that mimics multiple sclerosis. They found that the vaccine successfully halted the autoimmune reaction, preventing the immune system from attacking its own body.
In simpler terms, the inverse vaccine tricks the immune system by making it believe that the molecules it was wrongly attacking are actually friendly. It's like putting a "friendly" label on something the immune system thought was an enemy. This stops the immune system from causing harm and can potentially treat autoimmune diseases.
Impact: A Universal Approach to Autoimmune Disorders?
The groundbreaking nature of this research lies in its potential to serve as a foundational treatment for a myriad of autoimmune disorders. Central to this study is the innovative use of glycopolymer-conjugated antigens to induce tolerance in T cells, which are frequently implicated in autoimmune reactions. By specifically targeting these cells, there's a promising possibility that the treatment could be tailored for any disorder where T cells mistakenly assault the body's tissues.
What's particularly noteworthy is the treatment's ability to "reprogram" both memory and effector T cells—stages in T cell development that have historically been challenging to influence. These cells are often the primary contributors to the chronic inflammation and tissue damage seen in autoimmune diseases. Successfully inducing tolerance in these cells could revolutionize the treatment of conditions such as rheumatoid arthritis, lupus, and inflammatory bowel disease.
But the study's approach goes beyond mere suppression—it aims to retrain the immune system. This distinction suggests that the treatment might provide a lasting solution rather than just a temporary reprieve. Additionally, the antigen-specific nature of the therapy reduces the risk of making patients more susceptible to infections, a known drawback of many current immunosuppressive treatments.
Beyond autoimmune disorders, the potential applications of this research might extend to other immune dysfunctions. Allergies, for instance, which are often misunderstood as autoimmune disorders, are actually immune system reactions to foreign substances. If certain allergies are T-cell mediated, this strategy could potentially be applied to treat them as well.
In essence, this research lays the groundwork for a more precise, effective, and safer way to treat autoimmune diseases, potentially offering a one-size-fits-all solution that could be customized for various conditions.
Skeptical Analysis:
While the research presents a promising avenue for treating autoimmune disorders, there are several points that warrant a more skeptical lens.
Animal Models vs. Human Trials: The study's primary reliance on animal models raises questions about its applicability to humans. While animal studies provide valuable insights, human biology can be more complex, and results don't always translate directly.
Complexity of Autoimmune Disorders: The study's focus is on retraining T cells, but autoimmune disorders can involve a myriad of cellular mechanisms. For instance, B cells, another type of immune cell, are implicated in certain autoimmune diseases. It remains to be seen if this therapy would be as effective when B cells are the primary culprits.
Safety and Long-Term Effects: The potential long-term effects and side effects of using glycopolymer-conjugated antigens remain a concern. While no adverse effects were reported in the study, a longer observation period is crucial to ensure the therapy's safety in the long run.
Scalability and Cost: The production of specialized glycopolymer-conjugated antigens might pose challenges in terms of scalability and cost. This may be particularly challenging for autoimmune disease novelty and intrapatient antigen variation. While current immunosuppressive treatments have their drawbacks, they are widely available and often more affordable.
Conclusions and Next Steps:
The study presents a groundbreaking approach to treating autoimmune disorders by inducing tolerance in T cells. Using glycopolymer-conjugated antigens, the researchers were able to selectively target and "re-educate" T cells, reducing their aggressive response to self-antigens. This approach could revolutionize the way we treat a wide range of autoimmune disorders, from multiple sclerosis to rheumatoid arthritis, by offering a more targeted and less harmful alternative to current immunosuppressive therapies.
However, the journey from the lab to the clinic is a long one. Future research will need to focus on human trials to validate these findings in a more complex biological system. Additionally, the long-term safety and efficacy of this treatment need to be established. Researchers will also need to explore the therapy's applicability to autoimmune disorders that involve other types of immune cells, like B cells.
In the immediate future, we can expect to see more studies aimed at understanding the mechanisms behind this induced tolerance, as well as attempts to refine the glycopolymer technology for more effective and safer outcomes. The study opens the door to a new world of possibilities in treating autoimmune disorders, but there's still a long road ahead.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Pollard, A.J. and Bijker, E.M., 2021. A guide to vaccinology: from basic principles to new developments. Nature Reviews Immunology, 21(2), pp.83-100.
Tremain, A.C., Wallace, R.P., Lorentz, K.M., Thornley, T.B., Antane, J.T., Raczy, M.R., Reda, J.W., Alpar, A.T., Slezak, A.J., Watkins, E.A. and Maulloo, C.D., 2023. Synthetically glycosylated antigens for the antigen-specific suppression of established immune responses. Nature Biomedical Engineering, pp.1-14.
Williams, S.C.P., Inverse Vaccine Shows Potential to Treat Multiple Sclerosis and Other Autoimmune Diseases, University of Chicago.


Extremely interesting and valuable essay that is written in a way that makes the issues understandable to nonspecialists. Your essays on medical subjects are absolutely first rate, David. By all means continue in this vein. --Alex Klimoff
The reverse uno card on the body is the ultimate form of treatment I've learned. Very cool stuff as always David!