Donanemab: Alzheimer's Breakthrough or Just a Step?
A glance at the latest Phase 3 Trial and Its Implications for Alzheimer's Treatment
Introduction:
Alzheimer's disease remains one of the biggest unmet medical needs today, affecting millions worldwide. The disease is characterized by symptoms such as memory loss, confusion, and difficulty with speech and understanding; it's a disease that robs individuals of their cognitive functions and independence. According to the Alzheimer's Association, an estimated 6.7 million Americans (more than 50 million worldwide) aged 65 and older are living with Alzheimer's in 2023, a number expected to nearly double by 2050. Despite decades of research, there are still no treatments available that can significantly slow the progression of this devastating neurodegenerative disorder.
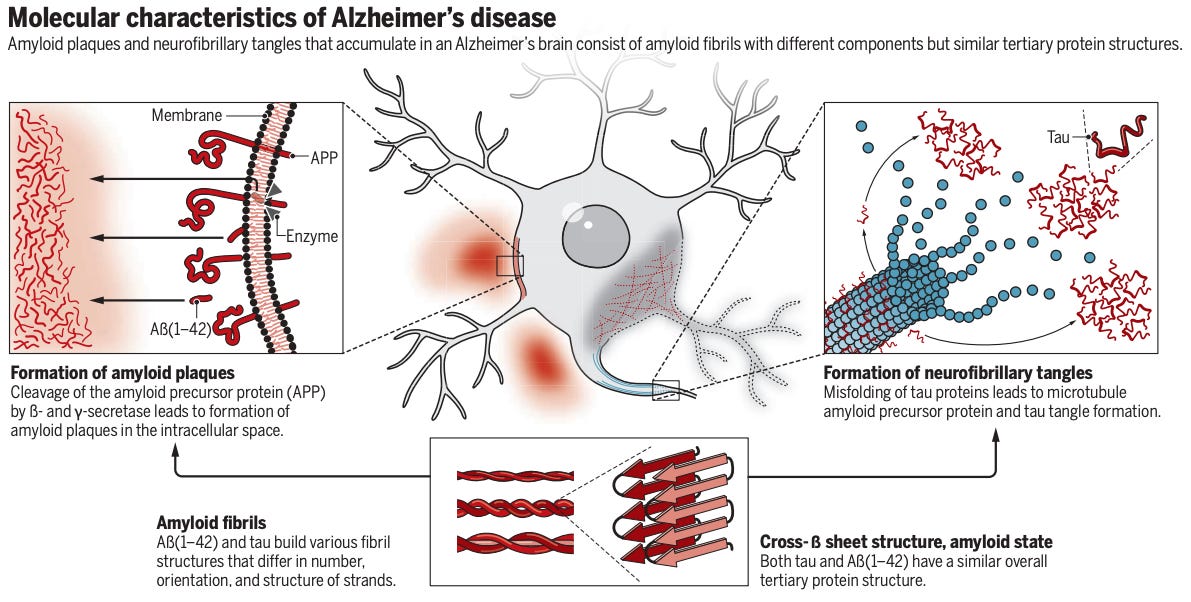
The underlying cause of Alzheimer's is still a matter of debate among researchers. The amyloid hypothesis, the dominant theory in Alzheimer's research, posits that the accumulation of amyloid plaques in the brain disrupt cell-to-cell communication (think of a clogged drain) between neurons. Researchers have been operating under the premise that removal of these plaques would alleviate these disease symptoms and potentially enable restored communication. This theory has guided much of the research and drug development over the past few decades, although it has also been the subject of intense scrutiny due to the failure of previous amyloid-targeting drugs (Herrup, 2015).
Enter donanemab, a drug being tested by Eli Lilly in a Phase 3 clinical trial. This antibody therapy is designed to clear amyloid plaques in the brain, offering a clear test of the amyloid hypothesis. Antibody therapies work by targeting specific molecules—in this case, amyloid plaques—to either block their activity or remove them altogether. This article aims to dissect the published Phase 3 study's findings, its potential impact on the field, and the questions that remain unanswered.
Claim:
Eli Lilly sponsored a Phase 3 clinical trial (recently published in JAMA) for an investigational Alzheimer's drug called donanemab (Sims, 2023). The conducted study was a double-blind, placebo-controlled, 76-week trial (1.5 years) that enrolled 1,736 individuals with early symptomatic Alzheimer's disease. The study screened and selected participants with amyloid plaques and tau tangles in their brains—two key pathological hallmarks of Alzheimer's.
Participants were randomly assigned to receive intravenous infusions of donanemab or a placebo every 4 weeks. Donanemab is an antibody therapy designed to clear amyloid plaque, a primary target in Alzheimer's drug development.
Remarkably, the trial met its primary goal: donanemab treatment significantly slowed decline on the integrated Alzheimer’s Disease Rating Scale (iADRS), a clinical rating scale that assesses both cognition and daily functioning. Compared to the placebo group, donanemab slowed progression by 35% in participants with early tau pathology and 23% in the overall trial population. Secondary outcomes, such as the Clinical Dementia Rating scale, also showed benefits.
Moreover, donanemab dramatically lowered amyloid plaque levels in the brain, with over 75% of treated participants reaching amyloid clearance based on PET scans. This not only provides evidence that the drug engages its target as intended but also marks a significant milestone in Alzheimer's research.
While not a cure, these clinically meaningful results suggest that donanemab may alter the course of early Alzheimer’s disease, allowing participants treated with the drug to remain healthier for longer compared to the placebo group.
Impact:
The results of this study have far-reaching implications, not just for Alzheimer's disease but potentially for the entire field of neurodegenerative disorders. The success of donanemab in clearing amyloid plaques opens up new avenues for targeted antibody therapies. For years, the 'amyloid hypothesis' has been a focal point in Alzheimer's research, but effective treatments have been elusive. This study provides compelling evidence that targeted antibody therapies can engage their intended targets and produce clinically meaningful outcomes.
Moreover, the impact of plaque removal on slowing disease progression is a significant breakthrough. If amyloid plaques are indeed a key factor in Alzheimer's progression, then therapies like donanemab could change the treatment paradigm for this devastating disease.
Additionally, the study's findings could have broader implications for other neurodegenerative diseases characterized by protein aggregation, such as Parkinson's and Huntington's disease. The use of antibodies for targeted treatment represents a paradigm shift from the more generalized approaches that have largely failed in the past.
Critical analysis:
The study hinges on the 'amyloid hypothesis,' the idea that the accumulation of amyloid plaques in the brain is the primary cause of Alzheimer's. While donanemab was certainly effective at removal of amyloid plaques, it has what seems to be a surprisingly mild reduction in Alzheimer's symptoms. Indeed, patients in the treatment group compared to the placebo had only a 22-35% reduction in disease progression. One would expect if the amyloid hypothesis the dominant factor in this disease for a larger effect or even a reversal of symptoms since the drug is nearly clearing the amyloid plaques. This weaker than expected effect clearly shows other mechanisms are at play. These observations (among other concerns) were similarly echoed by by JAMA editorial (Widera, 2023)
From a practical standpoint, the modest benefits of donanemab would be more compelling if the drug were low-risk, inexpensive, and easy to administer. However, this doesn’t seem to be the case. The study reported a notable incidence of brain swelling, known as amyloid-related imaging abnormalities, in 24% of participants treated with donanemab. While most cases were mild, some led to serious complications, underscoring the need for additional safety monitoring.
Lastly, the study does not address the financial and logistical challenges of administering such treatments. Antibody therapies like donanemab are often expensive and require intravenous administration, factors that could limit their accessibility and widespread use.
A final outstanding question I personally have is related to the drug’s delivery mechanism. One of the major challenges in treating neurological diseases is the blood-brain barrier, a protective shield that prevents substances (both good and bad) from entering the brain. This barrier is particularly selective when it comes to size; molecules larger than 500 Da typically struggle to cross it. Interestingly, the antibody donanemab is nearly 300 times larger than this upper size limit. So, how does it manage to bypass this biological barrier to reach the brain? This remains unclear in the article, although some of these details may be more obvious to others involved in the research.
Conclusions and next steps:
The donanemab study marks an important milestone in Alzheimer's research, demonstrating that targeted antibody therapies can have a clinically meaningful impact. With no other treatment alternatives available, this will provide some hope to many individuals and families. However, its clear this is just one piece of a larger puzzle. The study's modest success in slowing down Alzheimer's progression—while leaving many symptoms largely unaffected—underscores the need for a more comprehensive understanding of the disease's underlying mechanisms to inform new treatment strategies.
References:
Sims, J.R., Zimmer, J.A., Evans, C.D., Lu, M., Ardayfio, P., Sparks, J., Wessels, A.M., Shcherbinin, S., Wang, H., Nery, E.S.M. and Collins, E.C., 2023. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. Jama, 330(6), pp.512-527.
Herrup, K., 2015. The case for rejecting the amyloid cascade hypothesis. Nature neuroscience, 18(6), pp.794-799.
Alzheimer’s Association Website, (Accessed 06SEP2023), https://www.alz.org/alzheimers-dementia/facts-figures.
Pospich, S. and Raunser, S., 2017. The molecular basis of Alzheimer's plaques. Science, 358(6359), pp.45-46.
Widera, E.W., Brangman, S.A. and Chin, N.A., 2023. Ushering in a New Era of Alzheimer Disease Therapy. JAMA.



Neuro-degenerative diseases is one of my biggest fears in life of getting. Hopefully by the time I'm that age, it'll be as simple as a stomach ache to fix!