BioWire Weekly - 029
Biotech News
Happy Sunday Morning, BioWire Readers. I hope you had time to rest this weekend and are preparing to be relentless this week.
The topics this week:
Biotech and the Impact of Tariffs
New Engineered Gut Hormone Analogs Could Offer Adjunct Obesity Treatment
The Paradoxical Effect of Drug Shortages on Patient Accessibility and Pricing
The Machines are Coming—AI is Tackling Complex Tasks in Record Time
COVID Vaccine-Related Myocarditis in Young Adults
At that, let’s dive right in.
First, if you enjoy these updates, consider subscribing and becoming a part of our growing community!
Biotech and the Impact of Tariffs
Trade winds are blowing through the biotech world this week – and not the friendly kind. In early April, the U.S. enacted sweeping new tariffs across all sectors, and the industry is bracing for ripple effects from the lab bench to the manufacturing plant. While I’m far from an expert in economics, I thought it may be worth making some observations on how these policies may have a downstream effect, and quickly, in the biotech sector. For starters, biotech supply chains are globally entangled, with nearly 90% of U.S. biotechs relying on imported components for at least half of their products. Now, tariffs aimed at major trade partners (and expected retaliation in kind) are likely to raise the price of everything from raw reagents and lab consumables to specialized equipment. In short, the cost of medicine and science might climb overnight, and even routine shipments of cells or reagents could face delays.
Drug and biologics manufacturing could feel the pinch most acutely. Many companies partner with contract manufacturers in places like China or India to produce therapies, but over 60% of U.S. active pharmaceutical ingredients come from just those two countries. If those imports get caught in the tariff crossfire, firms may have to scramble for alternative suppliers or swallow higher production costs (likely a bit of both) that will be ultimately passed on to consumers. An industry survey already warned that 94% of biotech firms foresee higher manufacturing costs if tariffs hit key imports, and about half said they’d need to find new R&D or manufacturing partners – even delay some projects – in a tariff scenario. On the R&D side, labs depend on a free flow of materials and knowledge across borders. From high-end sequencers to genetically engineered cell lines, researchers often source critical tools globally. New trade barriers could mean waiting longer (and paying more) for that next-gen microscope or DNA synthesizer, and collaborations that once spanned oceans might hit bureaucratic snags.
Will these tariffs spur a push to “re-shore” biotech supply chains and reduce reliance on foreign suppliers? Perhaps eventually, with the continued development of AI and automation technologies, all of this will be possible and cheaper as we move into the future. But I think for now, we will have some choppy waters to navigate ahead.
New Engineered Gut Hormone Analogs Could Offer Adjunct Obesity Treatment
In another week about weight-loss drugs, researchers are exploring new hormonal pathways beyond the well-known GLP-1 receptor to provide additional therapeutic effects. Danish researchers (affiliated with biotech firm Gubra and Novo Nordisk) have been exploring peptide YY (PYY), a gut hormone that performs appetite suppression and satiety. PYY normally curbs appetite via the Y2 receptor in the brain, but its natural form produced in the body is short-lived, limiting any therapeutic application. In a hot-off-the-press study, published in Science Translational Medicine, researchers enhanced PYY to create long-acting analogs that only target the appetite-suppressing Y2 receptor, while avoiding related receptors (Y1, Y4, Y5) that can spur hunger or other undesirable side effects (Ostergaard et al., 2025)
How do you go about creating a molecule superior to what nature provides? The team combined good old-fashioned chemistry with cutting-edge AI. They systematically screened dozens of PYY variants, tweaking key amino acids to boost Y2 receptor activity and stability. This empirical screening was guided by structure-based design insights, including clues from AlphaFold, Google’s AI protein modeling tool, which the researchers used to predict how their peptide tweaks would fit into the Y2 receptor’s binding pocket. By modeling PYY’s interactions with Y2 (and the other Y receptors), they identified modifications that would increase specificity for Y2. They also tethered a small fatty molecule to the peptide to slow its breakdown, extending its half-life in the body. The result was a set of potent, Y2-only PYY analogs that stay active far longer than native PYY. It’s an impressive combination of rational design and AI-powered modeling (if you want to learn more about AI drug discovery, check out our previous article on In Silico Medicine).
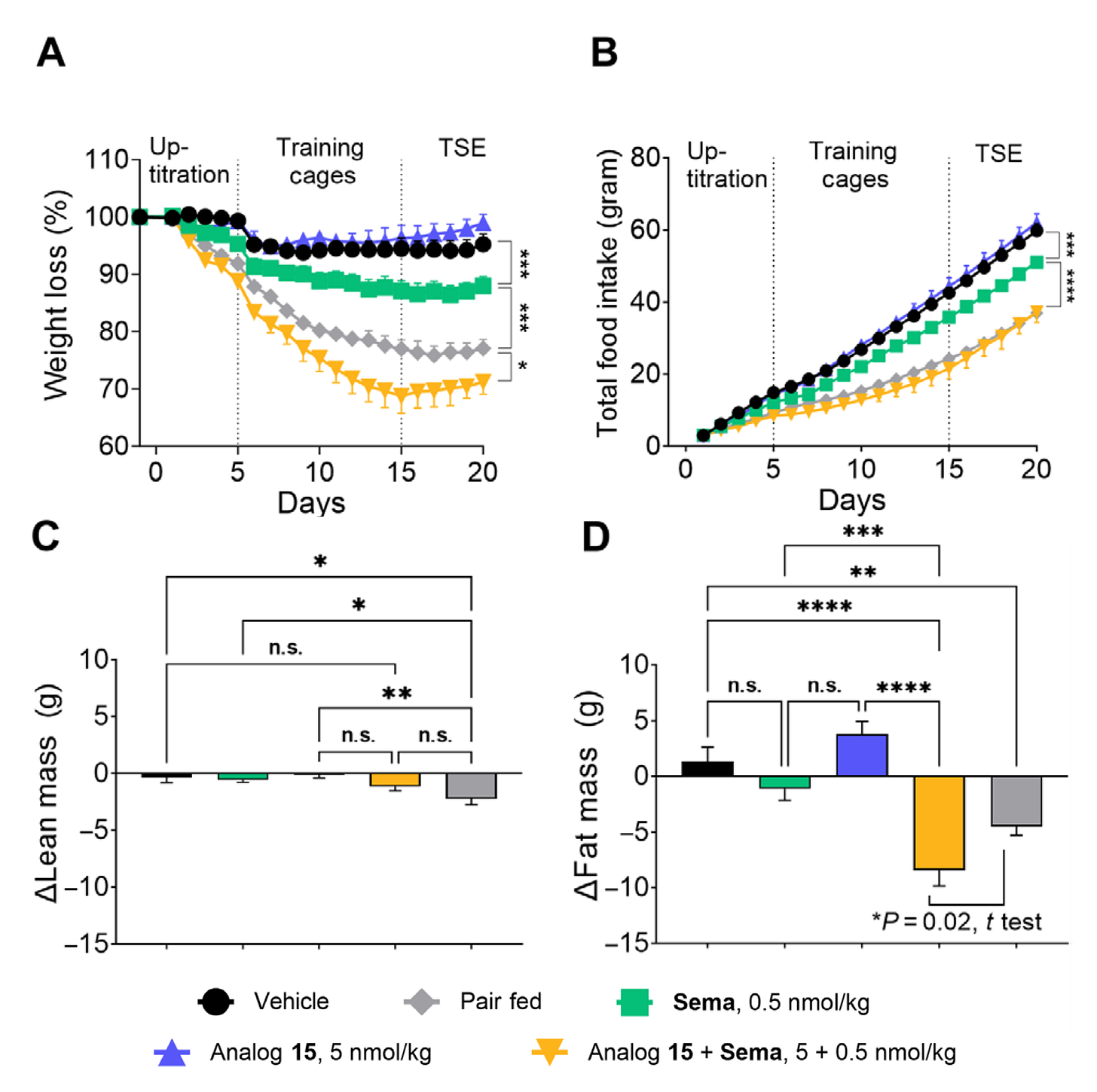
The laboratory protein engineering studies are cool, but now we’re getting to the really interesting stuff: do engineered PYY analogs help shed weight in preclinical animal models? The authors used a diet-induced obese mouse model to test their drugs. However, the initial results in these obese mice were underwhelming: even the best PYY analog on its own caused only modest weight loss (and I’d argue the data barely support even that claim). However, when the researchers combined their PYY analog with semaglutide (the active ingredient in Ozempic/Wegovy), things got a lot more interesting. In the obese mouse model, the combo led to a significantly greater weight loss than semaglutide or PYY alone, hinting at an additive, or even synergistic, effect. In diabetic animal models, adding PYY improved blood-glucose control beyond what GLP-1 therapy alone achieved. Notably, the extra pounds shed with the combo were almost entirely fat, with no significant muscle loss. Excited by these findings, the authors claim that these results lead to one of their analogs, PYY1875, to advance into a clinical trial for obesity.
I will admit, I was a bit skeptical of parts of this report. The casual mention of a clinical trial really piqued my interest though. If this combo were truly as impactful as the study claims, Novo Nordisk (and its shareholders) should be ecstatic. So I went searching for the trial, and that’s where things got weird. It turns out a Phase II trial of PYY1875 (in combination with semaglutide) had already been conducted - and quietly completed by early 2023! This 26-week, randomized, placebo-controlled study started in 2021, testing weekly PYY1875 at high (2.0 mg) or low (1.0 mg) doses on top of semaglutide 2.4 mg (the standard anti-obesity dose). By early 2023, the trial was finished. But what were the results? Strangely, nothing has been published or announced. It appears the PYY1875 program was halted, presumably due to a lack of the desired efficacy (or other strategic reasons).
Contrary to the paper published just two weeks ago, I can’t find any ongoing trials for PYY1875.
Why does this matter? If PYY1875’s fate has already been sealed (with no active clinical program), that’s vital context that should have been mentioned in the article, especially if it was published two years after the Phase II trial wrapped up. In addition, Science Translational Medicine is among the most prestigious academic journals and specifically focuses on preclinical insights with high translation potential as human therapies. It makes the omission of the human trial outcome even more unsettling. At a minimum, readers deserved to know that the “promising” PYY analog has already been tested in humans, with no reported success so far.
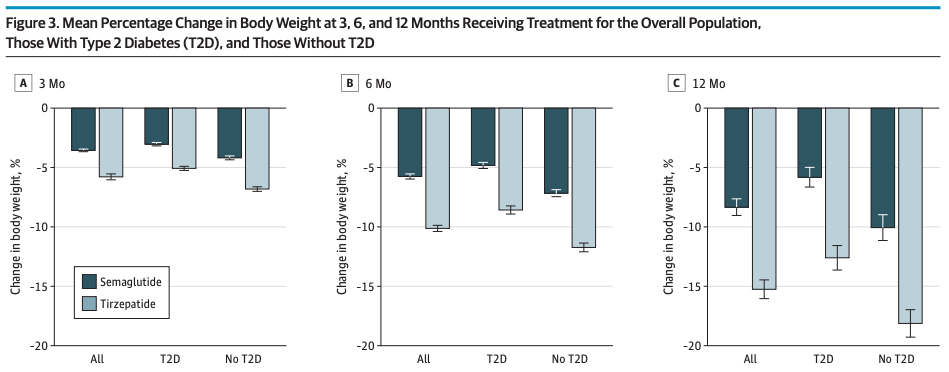
Setting aside the troubling publishing omission, the story is unfolding against the backdrop of a booming obesity drug market. It’s well established that obesity and its related illnesses drastically increase mortality and healthcare costs, and demand for effective weight-loss treatment is skyrocketing. The huge success of drugs like semiglutide (Novo’s GLP-1 receptor agonist) and tirzepatide (Eli Lilly’s dual GLP-1/GIP receptor agonist) has pharma companies racing to develop the next generation of therapeutics or competitors. Novo Nordisk, which funded the PYY project, is a major player in this race. However, recent trials indicate that semaglutide alone can’t quite match the efficacy of Lilly’s tirzepatide, likely because tirzepatide hits two hormonal targets (Rodriguez et al., 2024). Clearly, Novo is seeking an adjunct therapy to boost its GLP-1 treatment and reclaim best-in-class status without undermining its existing cash cow product. The PYY analog approach was one such attempt to amplify semaglutide’s effect by adding a second appetite-suppressing signal. Unfortunately, if the silent end of PYY1875 is any indication, not every hyped hormone analog will make it to the weight-loss wonder drug finish line.
The Paradoxical Effect of Drug Shortages on Patient Accessibility and Pricing
I have been consistently consistently tracking the weight-loss medication landscape, as you can see in the above section and prior editions of BioWire Weekly. At this point, I’ll assume some familiarity with GLP-1 receptor agonists, particularly blockbuster drugs semaglutide and tirzepatide. These drugs, marketed under brand names like Ozempic, Wegovy, and Mounjaro, are peptides that effectively aid in weight loss by curbing hunger and improving metabolism. While these drugs have been transformative for many, the medications typically run about $1,000 per month and are rarely covered by insurance. As a result, under normal circumstances, they would be financially out of reach for many Americans. Or at least, that's the case if you're buying directly from pharmaceutical giants like Novo Nordisk and Eli Lilly.
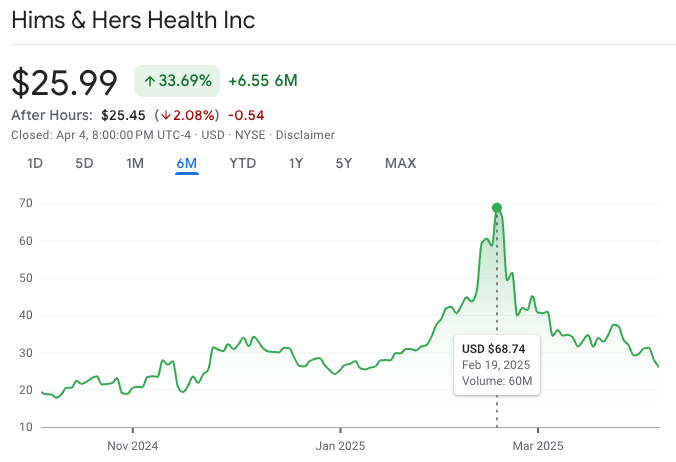
Here's where things get interesting. Due to ongoing drug shortages over the past three years, compounding pharmacies have exploited an FDA loophole to legally supply cheap, compounded versions of these peptides, sourced predominantly from China. These compounded versions are sold at a steep discount to eager consumers for around $200 per month (20% of the normal price). Now, over two million Americans have seized this opportunity, enabling unprecedented access to these effective but otherwise prohibitively expensive medications.
However, this loophole is quickly closing. In February, the FDA declared the semiglutide and tirzepatide shortages over, meaning compounding pharmacies and telehealth services (think Hims) must stop producing and selling these discounted GLP-1 formulations starting in late March for tirzepatide and late April for semaglutide. There is an interesting article written about this on Astral Codex Ten, looking at the implications. Overall, this has sparked creative (and desperate) medicolegal maneuvering among pharmacies and telehealth providers to maintain their market, ranging from minor dosage tweaks to novel formulations (think semaglutide gummies).
Novo Nordisk and Eli Lilly are countering with their own direct-to-consumer telehealth models, pricing medications around $500/month, higher than compounded versions but still below traditional retail. Moreover, Lilly has cleverly prevented dose-based arbitrage by offering single-dose vials without preservatives, ensuring consumers can't stretch their supply by buying higher potency vials and diluting.
Meanwhile, users on forums like Reddit are scrambling: some stockpiling medications, others resorting to DIY chemistry by mixing peptides sourced from overseas suppliers, risky territory fraught with potential dosing errors or contamination risks.
Ultimately, this scenario is a morbidly fascinating, if unintended, experiment in semi-free-market medicine. Despite minimal regulation and oversight, patients thrived on these cheap compounded peptides. Anyone who cares about public health should find it interesting that adult obesity rates in the US dropped for the first time in our lifetime in 2023 and likely 2024. Maybe it’s time to challenge some of these regulatory structures and patent protections.
The Machines are Coming—AI is Tackling Complex Tasks in Record Time
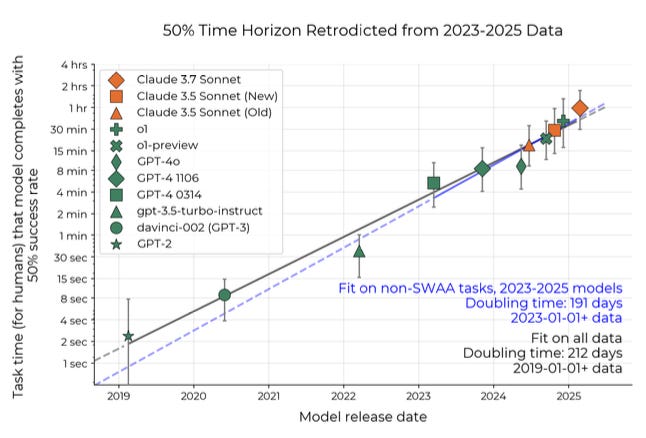
How long of a project can today’s AI really handle compared to a human? A new study from the Model Evaluation & Threat Research (METR) team tries to answer question in a new preprint article (Kwa et al., 2025), with a metric called the “task-completion time horizon.” In simple terms, this is the length of time a task takes a person to finish when an AI can achieve the same task with 50% success. Researchers first timed human experts on a battery of coding, security, and reasoning tasks to set a baseline. The time-horizon metric matters because it translates AI capability into human terms – an intuitive gauge of how close AI is getting to tackling real-world projects (not just bite-sized benchmarks) at a human-like level.
The findings are both impressive and a bit startling. Back in 2019, OpenAI’s GPT-2 (an early large language model) failed to complete any task that took human experts more than about one minute. Fast-forward to today: Anthropic’s latest model Claude 3.7 can reliably solve tasks that require roughly 50 minutes of human work with a 50% success rate. In other words, its 50%-completion time horizon is nearly an hour – a remarkable leap in a few years. In fact, this “AI time horizon” has been growing exponentially, roughly doubling every seven months since 2019. The trend even appeared to accelerate in 2024, with cutting-edge models extending their reach at an even faster clip. Extrapolate this curve out a bit, and you can imagine that by the late 2020s (around 2029), AI systems might handle projects that take humans a full month of work, at least half the time.
Why does jumping from one minute to one hour matter? Because a month of dedicated human expertise is enough to found a startup or make a scientific discovery. The prospect of AI reaching that level in just a few years is both thrilling and startling. It suggests we’re on the cusp of AI that can autonomously carry out multi-week projects (in potentially a fraction of the time) – potentially a game-changer for productivity. But some experts urge caution about reading too much into these forecasts. It’s tempting to draw a straight line and predict an “AI does month-long tasks” future, but there’s a lot we don’t yet know about how these models will actually be used or what bottlenecks might appear. Raw capability is one thing; translating it into real-world impact (or avoiding unintended consequences) is another. In short, the time-horizon metric is insightful, but it’s not a crystal ball.
The new metric itself is a clever way to track meaningful progress, yet it has limitations. By design, it evaluates tasks that are similar to what the AI was trained on – so it might not reveal how well a model generalizes to truly novel challenges outside that comfort zone. The METR team acknowledges that their tasks (even if drawn from real work scenarios) can’t capture all the messiness of actual large-scale projects. They did check that the trend holds even when tasks resemble real-world jobs, but it’s still an imperfect proxy. There are also practical constraints to consider: the past five years of AI gains were supercharged by ever-larger models and more computing power, and that can't continue indefinitely due to physical and economic limits. Future progress might slow if we hit those walls. On the other hand, improvements in algorithms – and giving AI models more “agency” to self-correct and manage longer processes – could keep pushing the frontier. The researchers suggest the next breakthroughs may come from pairing AIs with humans, combining machine efficiency with human judgment to tackle big tasks. As AI systems inch toward handling weeks-long or month-long projects, we face a double-edged sword: incredible new capabilities and productivity on one side, and on the other, serious questions about safety and oversight when machines start working on complex tasks with minimal human intervention. The clock is ticking, and the horizon is getting ever longer.
If you had such a model at your fingertips, capable of thinking and working on a problem that would take a team of humans months, what would you send it to tackle? Leave your thoughts in the comments.
COVID Vaccine-Related Myocarditis in Young Adults
Even years on, COVID-19 and mRNA vaccines remain politically charged topics, making objective data a rare commodity. So, a new retrospective study published in JAMA caught my eye by shedding some light on the long-term outcomes of vaccine-related myocarditis. Researchers analyzed data from 4,635 French patients hospitalized for myocarditis, comparing cases linked to mRNA vaccines, COVID infections, and conventional (non-COVID related) causes.
The headline finding: vaccine-induced myocarditis, though serious enough to warrant hospitalization, had notably better long-term outcomes compared to myocarditis triggered by COVID-19 infection or conventional causes. After an 18-month follow-up, mortality and severe cardiovascular complications were significantly lower among those whose myocarditis followed vaccination. Only 0.2% of vaccine-associated patients died compared to approximately 1.3% in the other two groups, and vaccine-related cases were less likely to require readmission or intensive interventions like heart transplants or mechanical support.
However, there’s an important nuance the authors didn’t highlight clearly: myocarditis risk was dose-dependent, primarily affecting younger men after their second mRNA vaccine dose. Despite a good long-term prognosis, affected individuals still required hospitalization and months of follow-up care.
Notably, the authors refrained from discussing vaccine policy implications or recommendations, leaving the broader debate unresolved. Still, their findings suggest that while vaccine-induced myocarditis presents a genuine risk, particularly for younger males receiving multiple doses, its long-term outcomes are typically less severe than those of myocarditis from COVID itself or other common causes.
These newsletters take significant effort to put together and are totally for the reader's benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Kwa, T., West, B., Becker, J., Deng, A., Garcia, K., Hasin, M., Jawhar, S., Kinniment, M., Rush, N., Von Arx, S. and Bloom, R., 2025. Measuring AI Ability to Complete Long Tasks. arXiv preprint arXiv:2503.14499.
Østergaard, S., Jessen, C., Paulsson, J.F., Kasimova, M.A., Conde-Frieboes, K.W., Straarup, E.M., Skyggebjerg, R.B., Ynddal, L., Sanfridson, A., Wulff, B.S. and Chambers, A.P., 2025. Variant screening of PYY3–36 leads to potent long-acting PYY analogs with superior Y2 receptor selectivity. Science Translational Medicine, 17(791), p.eadq6392.
Rodriguez PJ, Cartwright BM, Gratzl S, Brar R, Baker C, Gluckman TJ, Stucky NL. Semaglutide vs tirzepatide for weight loss in adults with overweight or obesity. JAMA internal medicine. 2024 Sep 1;184(9):1056-64.
Semenzato, L., Le Vu, S., Botton, J., Bertrand, M., Jabagi, M.J., Drouin, J., Cuenot, F., Zores, F., Dray-Spira, R., Weill, A. and Zureik, M., 2024. Long-term prognosis of patients with myocarditis attributed to COVID-19 mRNA vaccination, SARS-CoV-2 infection, or conventional etiologies. JAMA, 332(16), pp.1367-1377.
Yang, C. and Tebbutt, S.J., 2025. Long-Term Prognosis of Patients With Myocarditis. JAMA, 333(8), pp.720-721.
https://www.bio.org/press-release/new-survey-us-biotechs-warn-tariffs-could-impede-access-cures-stifle-innovation#:~:text=findings%2C%20nearly%2090,European%20Union%2C%20China%2C%20and%20Canada
https://www.wilsoncenter.org/article/strengthening-us-mexico-quality-pharmaceutical-supply-chains#:~:text=Despite%20current%20interest%20in%20medicines,Notably







I talked to my buddy who’s about to take his LSAT and he is currently working as a paralegal. He basically uses AI for every single email, form of communication, and legal document and nobody notices. The legal field is about to be set on fire once the younger more techs-savvy people replace the current old guard.