BioWire Weekly - 025
Biotech News
Happy Sunday, Readers.
I usually start by telling you all to be relentless. But this week, I hope you’re all taking some time to recharge, see family, and enjoy your Christmas Holiday.
Some quick housekeeping before we start—I can’t believe we’re already in the final weeks of 2024. It’s been a wild year, and keeping up with news in Tech and Biotech has felt like drinking from a fire hose. I’m hoping to squeeze in a few more articles before the end of the year, but holiday travel might make that tricky.
I also want to give a quick update on the stack, which will come in a longer post as well. We’ve seen tremendous growth in readership this year. Just as a hint, we’re now read in 53 countries—including some I would’ve struggled to point to on a map a year ago. That’s absolutely humbling, and I’m so grateful for all of you.
With that out of the way, let’s dive into this week’s biotech news.
Clinical Studies of AI-Discovered Drugs are Rapidly Growing
Technical Report on Mirror Bacteria Highlights Dangers and Risks of Synthetic Biology
AI Physicians are on the Horizon
FDA Cracks Down on Unapproved GLP-1 Drugs for Weight Loss
Clinical Studies of AI-Discovered Drugs are Rapidly Growing
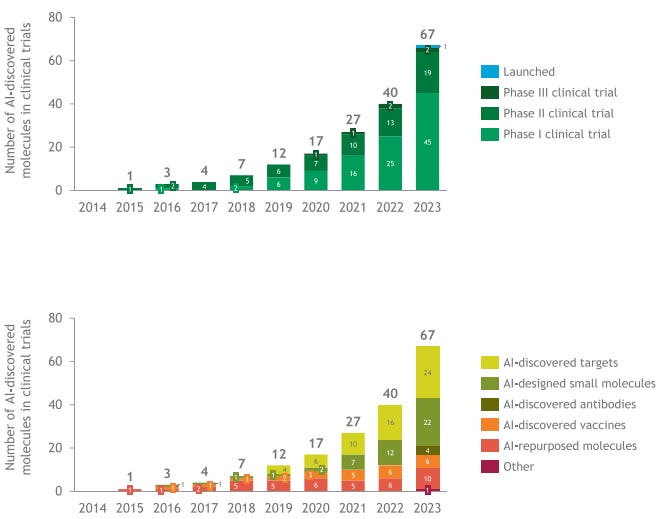
Earlier this year, I wrote an article about why I believe artificial intelligence will revolutionize biotechnology, particularly in drug discovery. However, the field of AI-powered drug discovery is in its infancy. With all this bullishness, it is worth asking: are there any early indicators of well AI-designed drugs perform once they reach clinical trials? Fortunately, a recent analysis published in Drug Discovery Today reveals promising results, offering both hope and lessons for the future of AI-powered drug development (Jayatunga et al., 2024).
The Boston Consulting Group analyzed clinical trial outcomes from 75 AI-discovered drugs developed by AI-native biotech companies, most of which are partnered with pharmaceutical giants. The findings are particularly striking at the Phase I stage, where AI-discovered molecules demonstrated an 80–90% success rate. This far exceeds traditional Phase I averages of 40–65%, suggesting AI models are adept at identifying drug candidates with favorable safety and pharmacokinetic profiles.
In Phase II, where proof-of-concept and efficacy are tested, success rates drop to 40%, aligning closely with historical averages. The study notes that the "hard problem" of translating AI-predicted biology into clinical efficacy remains. However, a deeper dive reveals that only a fraction of Phase II failures were due to negative outcomes—many were discontinued for business or operational reasons, reflecting broader economic challenges associated with biotech.
The success of AI in Phase I trials could stem from its ability to optimize drug-likeness, predict safety, and explore novel chemical space, reducing early failures. Notably, AI-driven approaches are diversifying. While AI-repurposed drugs once dominated, AI-discovered small molecules, vaccines, and biologics are gaining ground, with oncology leading as the most common therapeutic focus.
The authors propose an optimistic thought experiment: if AI’s early success rates hold and Phase III remains on par with traditional averages, end-to-end clinical trial success rates could nearly double from 5–10% to 9–18%. This would significantly enhance R&D productivity, enabling faster, cheaper delivery of innovative medicines to patients.
Technical Report on Mirror Bacteria Highlights Dangers and Risks of Synthetic Biology
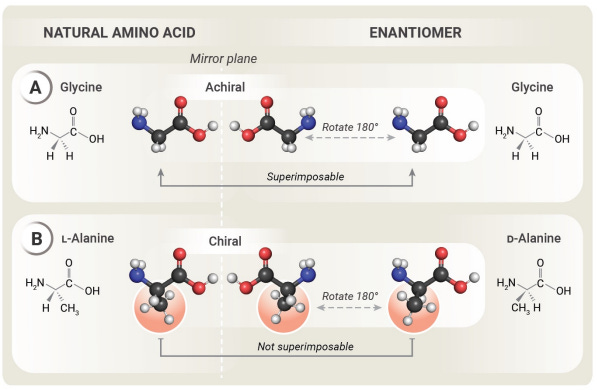
The concept of "mirrored life" has emerged as a fascinating but potentially dangerous frontier in synthetic biology, according to a recent report. Mirrored life refers to organisms built with reversed chirality—the flipped molecular structure of proteins and nucleic acids compared to natural life. For those unfamiliar with the concept of chirality, think of your left and your right hands, they are non-superimposable mirror images of each other. A practical example is found in the sugars we consume for energy. While right-handed sugars are metabolized by our bodies for fuel, left-handed sugars are not, allowing us to enjoy their sweetness without the added calories. While they have interesting opportunities for breakthroughs like novel drug manufacturing and enzyme engineering, these mirror organisms also present unique and significant risks.
One of the most concerning aspects of mirror life is its potential resistance to natural biological checks and balances. These organisms would likely be undetectable by natural immune systems and immune to conventional antibiotics, as their molecular structures are fundamentally different from existing pathogens. If accidentally released, mirrored organisms could proliferate unchecked, causing ecological and medical crises without facing natural predators or immune responses.
The report believes that the development of fully functional mirrored life remains a unfeasible in the near future, but progress in synthesizing mirrored biomolecules has already sparked heated debate. Critics argue that the risks far outweigh the rewards, with calls for international regulations to prevent accidents or misuse. Proponents, however, contend that mirrored life could lead to transformative innovations in medicine and technology.
As this field progresses, the question remains: can we ensure the safe development of mirrored life, or do the risks necessitate preemptive restrictions? This pivotal debate highlights the double-edged sword of synthetic biology—immense potential tempered by profound dangers. This all goes without even mentioning viral research centers studying dangerous pathogens and the likelihood they are the originators of a certain pandemic that changed the world in 2020…
AI Physicians are on the Horizon
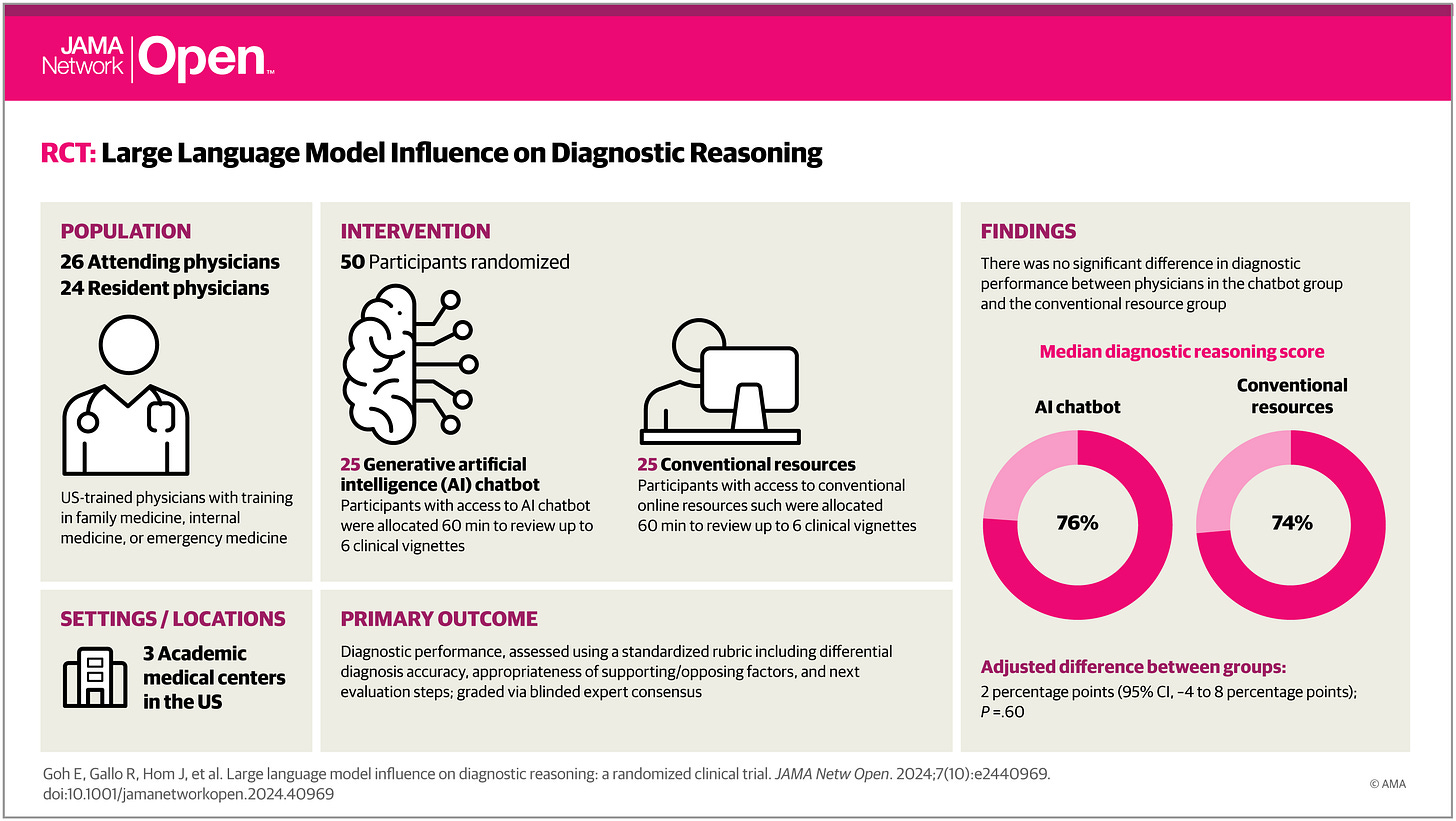
A recent randomized clinical trial recently explored the role of large language models (LLMs) like GPT-4 in enhancing physician diagnostic reasoning. The findings, published in JAMA Network Open, present a snapshot of the current capabilities and limitations of AI and clinician pairings in the clinical settings (Goh et al., 2024).
In the trial, 50 physicians from internal, family, and emergency medicine specialties were randomized into two groups: one with access to GPT-4 alongside conventional diagnostic tools and the other relying solely on traditional resources. Both groups evaluated six clinical vignettes, or patient related case scenarios, designed to simulate complex real-world cases. Surprisingly, the presence of GPT-4 did not significantly improve diagnostic performance. Physicians using GPT-4 scored a median of 76% on diagnostic reasoning tasks, marginally higher than the 74% achieved by those using only conventional resources. However, the difference in performance did not reach statistical significance.
In a surprising twist, GPT-4’s standalone diagnostic performance outshone both groups, achieving a median score of 92%. This suggests that the model’s diagnostic capabilities are strong, but physicians may not yet be equipped to fully integrate its insights into their decision-making processes. The authors noted that structured training in prompt engineering and human-AI interaction could help clinicians harness the full potential of tools like GPT-4. Alternatively, this result may hint at the possibility that in certain aspects of diagnostics, AI systems are already capable of surpassing human expertise.
Another key insight was the gap in time efficiency. Physicians using GPT-4 completed cases slightly faster, but the 82-second reduction per case was not statistically significant. While this hints at the potential of LLMs to streamline workflows, it also shows that they have not yet achieved transformative efficiency gains.
Overall, the study underscores the importance of deliberate integration of LLMs into clinical practice. Training clinicians to use AI tools, standardizing prompts, and designing systems that facilitate collaboration between humans and machines could unlock their full potential. While GPT-4’s standalone diagnostic accuracy is impressive, its true value lies in complementing—not replacing—human expertise.
FDA Cracks Down on Unapproved GLP-1 Drugs for Weight Loss
GLP-1 receptor agonists have soared in popularity over recent years due to their proven effectiveness in weight loss and diabetes management. This surge in demand has caused significant shortages, prompting compounding pharmacies to step in and fill the gap. However, the FDA has raised serious concerns about the growing availability of unapproved and compounded versions of these drugs, including semaglutide and tirzepatide. While FDA-approved medications like Ozempic, Wegovy, and Mounjaro are widely prescribed, these unregulated alternatives are becoming increasingly common and there is concern of serious health risks.
What sets compounded drugs apart? Unlike FDA-approved medications, compounded drugs are custom-made by mixing ingredients to address specific patient needs or general shortages. However, they bypass the FDA's safety and efficacy evaluations. This lack of oversight and has led to reports of adverse effects, including dosing errors and hospitalizations. According to the report, these compounded versions may also contain inconsistent dosages or unverified ingredients, increasing the risks to patients.
Beyond compounding pharmacies, even less scrupulous vendors have entered the market, selling counterfeit GLP-1 products. The FDA is actively cracking down on these companies, many of which market their products as “research use only” but sell them for off-label human use. Recent warning letters have targeted online vendors contributing to this growing trend, fueled by the surging popularity of GLP-1 therapies.
What does this all mean? The extraordinary demand for weight-loss products like GLP-1 agonists is far outpacing supply, leading patients to seek alternative—and often riskier—options at lower costs. My advice to anyone seeking these drugs is simple: consult licensed healthcare providers and obtain medications from trusted, state-licensed pharmacies. Anything less involves significant and unnecessary risk.
References:
Goh, E., Gallo, R., Hom, J., Strong, E., Weng, Y., Kerman, H., Cool, J.A., Kanjee, Z., Parsons, A.S., Ahuja, N. and Horvitz, E., 2024. Large language model influence on diagnostic reasoning: a randomized clinical trial. JAMA Network Open, 7(10), pp.e2440969-e2440969.
Jayatunga, M.K., Ayers, M., Bruens, L., Jayanth, D. and Meier, C., 2024. How successful are AI-discovered drugs in clinical trials? A first analysis and emerging lessons. Drug Discovery Today, p.104009.
https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fdas-concerns-unapproved-glp-1-drugs-used-weight-loss
https://purl.stanford.edu/cv716pj4036





AI will replace software engineers first, mark my words!