BioWire Weekly - 012
Biotech News
Happy Friday’s Eve, Readers.
In this week’s BioWire, we dive into some exciting developments in biotech, including the successful phase 2 trial of a bacteriophage therapy for multidrug-resistant infections, Neuralink’s latest milestones in brain-computer interfaces, DePuy’s innovative robotic spine surgery system, and a promising new blood test for early Alzheimer’s detection. Plus, we’ll explore a novel treatment for dry eye disease that’s making waves in clinical trials. Let’s get started.
Locus Biosciences Successful Phase II Part I
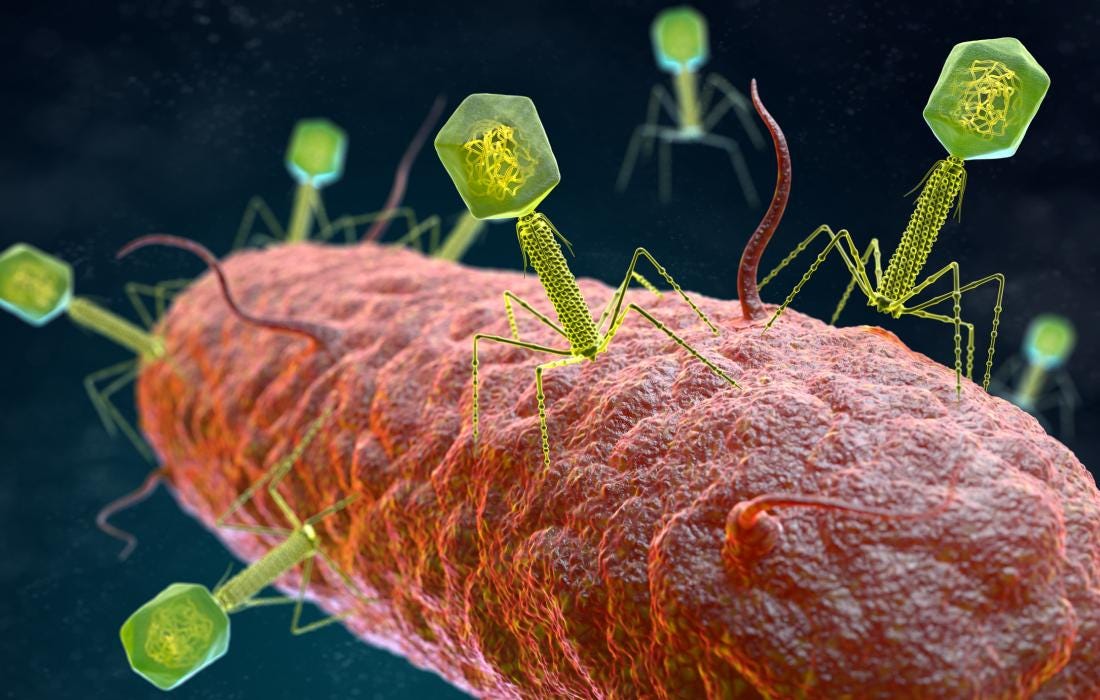
Locus Biosciences has reported encouraging results from Part 1 of its Phase 2 ELIMINATE trial, which is testing LBP-EC01, a CRISPR-enhanced bacteriophage therapy designed to treat urinary tract infections (UTIs) caused by antibiotic-resistant E. coli. Bacteriophages are viruses that specifically target and destroy bacteria, making them a promising tool against antibiotic-resistant infections. This trial is particularly significant as it targets multidrug-resistant (MDR) pathogens, a growing global health concern.
The open-label, randomized trial enrolled 39 adult female patients across six clinical centers in the U.S. The study’s primary and secondary objectives were successfully met, demonstrating that LBP-EC01 was well-tolerated and achieved high drug exposure at the site of infection. These promising results which were recently published in The Lancet (Kim et al, 2024).
Part 2 of the ELIMINATE trial has commenced, focusing on a larger, randomized, controlled, double-blind study to further evaluate the efficacy, safety, and long-term impact of LBP-EC01. This study is backed by the Biomedical Advanced Research and Development Authority (BARDA) and represents a significant milestone in developing new treatments for antibiotic-resistant infections. I plan to give a more indepth review on bacteriophage and these published results in an upcoming article.
Neuralink developments
Imagine you are at the beach with friends on a hot, sunny day. You dive into the water to cool off, but instead of feeling the expected splash, something hard strikes your head. You black out, and when you come to, you realize you can’t move. To your friends horror, you appear to be floating face down, twitching in the water. They pull you to safety, and after emergency surgery, you’re told you’ve suffered a spinal cord injury and face a long, uncertain road to recovery. Despite years of rehabilitation, you remain paralyzed from the neck down. This is the story of Nolan, Neuralink’s first patient.
Neuralink is testing an experimental brain-computer interface called ‘Telepathy.’ This device, with 64 threads covered in electrodes, is surgically implanted into the brain and calibrated to allow quadriplegics like Nolan to interact with their computers—or potentially other electronic devices—simply through thought. We’ve discussed Neuralink in previous posts, but I recently came across an interview with Lex Fridman featuring key figures from the Neuralink team, including Elon Musk, COO DJ Seo, Head Neurosurgeon Matthew MacDougall, and Nolan Arbaugh himself. The interview is over 8 hours long, but it’s worth a listen.
It’s hard to believe it’s already been 7 months since Nolan (patient 1) received his Neuralink device. As we discussed, this came with some challenges, such as the retraction of threads containing electrodes. Despite these setbacks, Nolan appears to be extremely happy with the device. Now, Neuralink has announced they have enrolled a second patient, who has already undergone the procedure. Judging by the 7-month gap between patient 1 and patient 2, I would wager there have been some changes made to overcome the issues in the first procedure. While details are scarce, the initial procedure reportedly went well. Elon Musk mentioned during the podcast that Neuralink aims to enroll 10 patients by the end of the year, but they’ll need to accelerate their efforts to meet that goal.
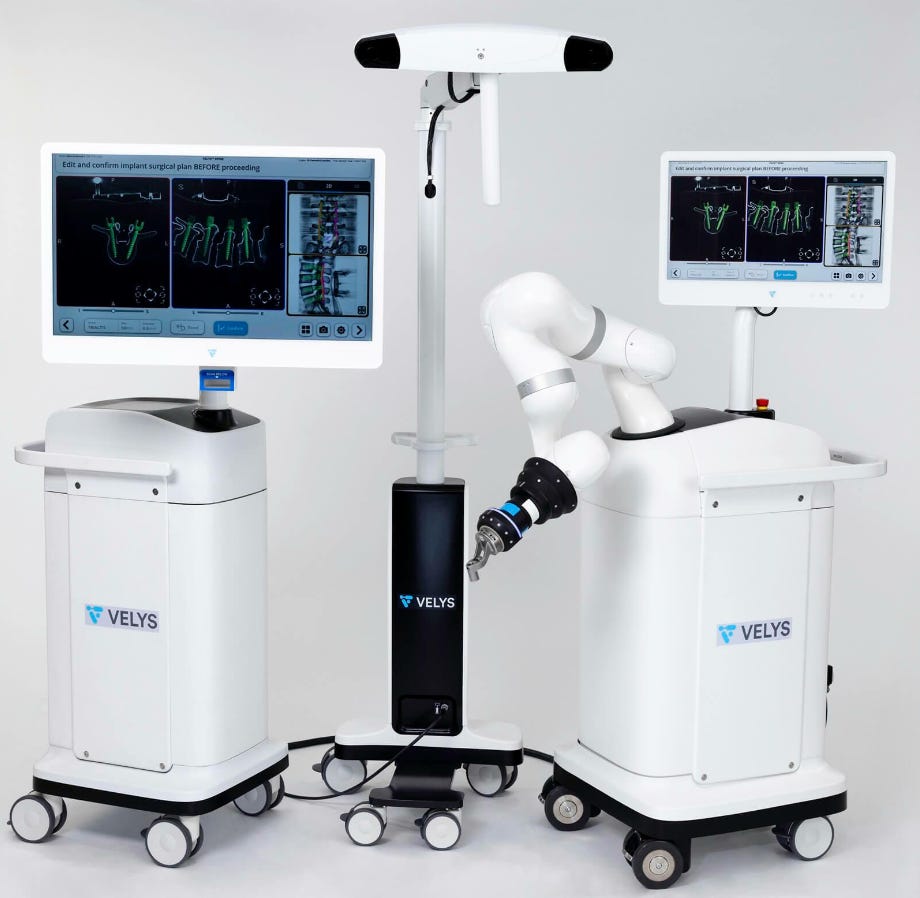
DePuy Launches New Robotic Spine Surgery Program
DePuy Synthes, a company under Johnson & Johnson MedTech, has launched the VELYS™ Active Robotic-Assisted System, aimed at enhancing spinal surgery. This system is designed to aid surgeons in performing complex spinal fusion procedures by integrating advanced robotic assistance with precise navigation tools. The developers tout the dual-use platform’s ability to offer flexibility, allowing surgeons to tailor their approach based on individual patient needs. With this innovation, DePuy Synthes is looking to set a new standard in spinal surgery, focusing on improving patient outcomes and advancing the capabilities of spine surgeons. The VELYS™ system will be part of the broader VELYS™ Enabling Technologies Portfolio, integrating with existing Johnson & Johnson MedTech spine products, and is expected to be commercially available in the first half of 2025. This launch appears to be part of a larger commitment to leveraging digital advancements to enhance surgical care.
Blood Biomarkers for Early Detection of Alzheimer Disease
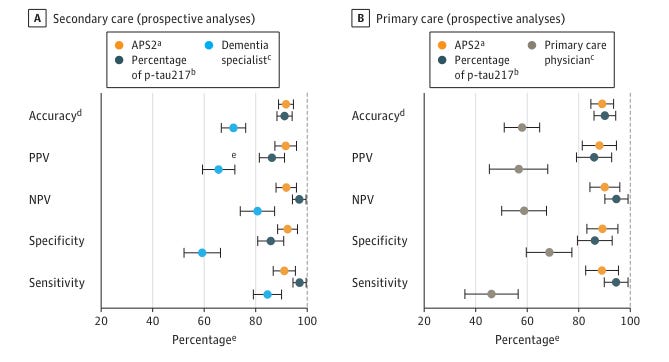
Alzheimer's disease (AD) remains a significant challenge in early diagnosis and effective treatment, but recent advancements have introduced a promising blood test that measures key proteins associated with AD pathology (Palmqvist et al, 2024). The test focuses on plasma phosphorylated tau 217 (p-tau217) and amyloid-beta (Aβ) ratios, two proteins that play a critical role in the disease's progression by forming neurofibrillary tangles and amyloid plaques in the brain, respectively. By combining these measures into an amyloid probability score (APS2), the test achieves high diagnostic accuracy (88%-92%) across both primary and secondary care settings. This innovation marks a substantial step forward, potentially transforming how AD is diagnosed by enabling earlier detection and more precise treatment strategies. The implications are particularly significant given the limitations of current diagnostic methods, which often miss early signs of the disease, especially in primary care settings where most patients are first evaluated.
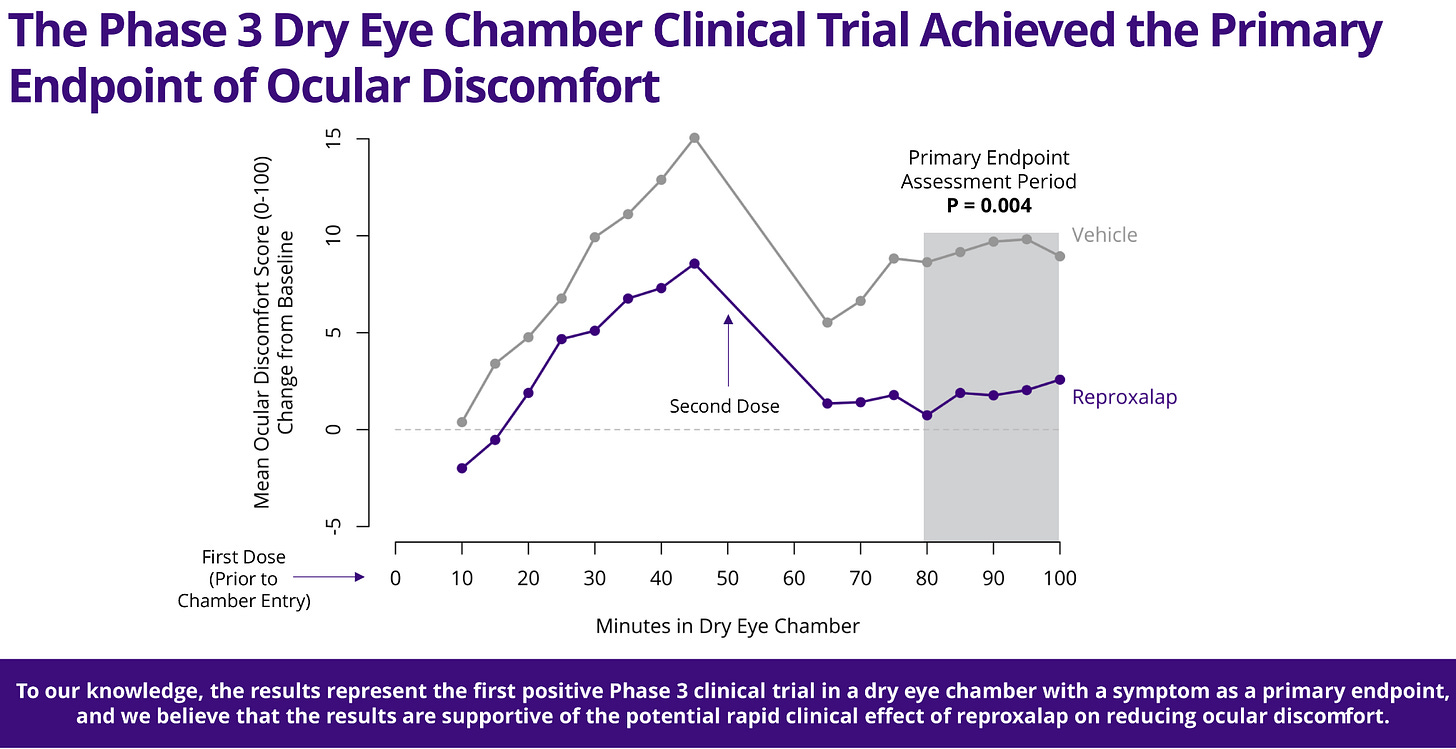
A new drug for dry eye disease
Aldeyra Therapeutics is preparing to refile with the FDA after its dry eye drug, reproxalap, showed promising results in a new phase 3 clinical trial. Reproxalap is an investigational small-molecule drug that targets ocular discomfort by addressing the underlying inflammation that causes the discomfort. It works by neutralizing reactive aldehydes—byproducts of oxidative stress that contribute to inflammation and irritation in the eyes. This novel approach demonstrated rapid effectiveness in a controlled dry eye chamber, potentially offering more reliable relief for patients. Aldeyra plans to resubmit for FDA approval later this year, while also exploring other potential uses for the drug, such as treating allergic conjunctivitis.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References
Kim, P., Sanchez, A.M., Penke, T.J., Tuson, H.H., Kime, J.C., McKee, R.W., Slone, W.L., Conley, N.R., McMillan, L.J., Prybol, C.J. and Garofolo, P.M., 2024. Safety, pharmacokinetics, and pharmacodynamics of LBP-EC01, a CRISPR-Cas3-enhanced bacteriophage cocktail, in uncomplicated urinary tract infections due to Escherichia coli (ELIMINATE): the randomised, open-label, first part of a two-part phase 2 trial. The Lancet Infectious Diseases.
Palmqvist, S., Tideman, P., Mattsson-Carlgren, N., Schindler, S.E., Smith, R., Ossenkoppele, R., Calling, S., West, T., Monane, M., Verghese, P.B. and Braunstein, J.B., 2024. Blood Biomarkers to Detect Alzheimer Disease in Primary Care and Secondary Care. JAMA.
https://www.jnjmedtech.com/en-US/news-events/depuy-synthes-launches-its-first-active-spine-robotics-and-navigation-platform
https://www.businesswire.com/news/home/20240808408806/en/Aldeyra-Therapeutics-Achieves-Primary-Endpoint-in-Phase-3-Dry-Eye-Disease-Clinical-Trial-of-Reproxalap