BioWire Bytes 023 – Scientists Restarted Dead Brains. Things Got Weird.
Dead Pig Brains Resumed Cellular Function Four Hours After Death
A surgeon is performing a heart transplant. He cracks the chest, connects the patient to bypass, and removes the failing heart. He sutures in the donor organ. Aorta to aorta. Pulmonary artery. Vena cava. Left atrial cuff. He removes the cross-clamp and rewarms the heart with perfused blood. Nothing. He defibrillates. Nothing. Inotropes. Mechanical support. Six hours later, the team withdraws care. But when did the patient actually die? Was it when the old heart stopped? When the new one never started? If it had resumed rhythm ten minutes after they quit, would the patient have been alive the whole time?
The medical establishment treats brain death as binary. Circulation stops. Neurons lose oxygen. Within minutes, cells enter irreversible decline. That timeline underpins the legal definition of death, organ procurement windows, and the clinical default that a post-mortem brain is biologically inert. In 2019, Nenad Sestan’s lab at Yale tested that assumption directly, and the results don’t support it.
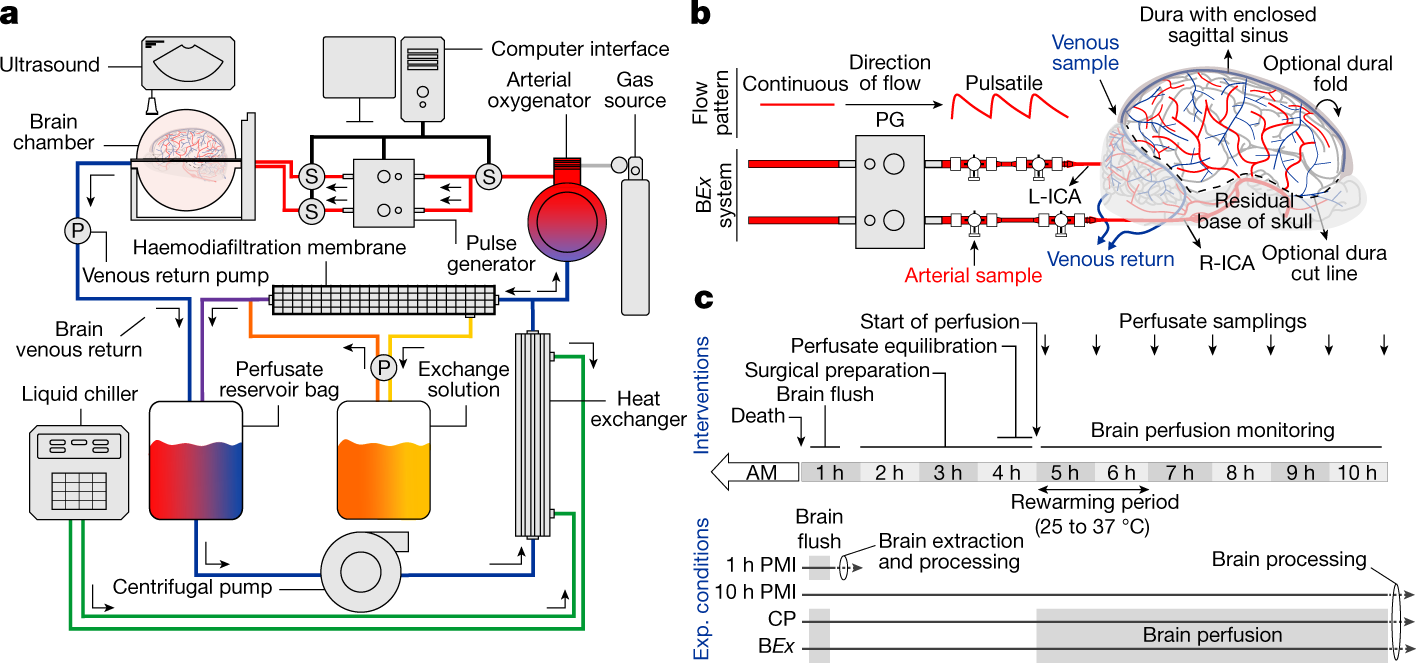
Vrselja and colleagues took 32 pig brains from a slaughterhouse and waited four hours after death. They connected them to BrainEx, a perfusion system that pushes synthetic oxygenated solution through the brain’s own vasculature. The perfusate carried cytoprotective agents, anti-inflammatories, and neural activity blockers. Control brains received no perfusion or perfusion without the active solution. Six hours later, the treated brains were compared against both control groups (Vrselja et al., 2019).
Figure 1. BrainEx perfusion system and experimental workflow. Adapted from Vrselja et al., Nature 2019.
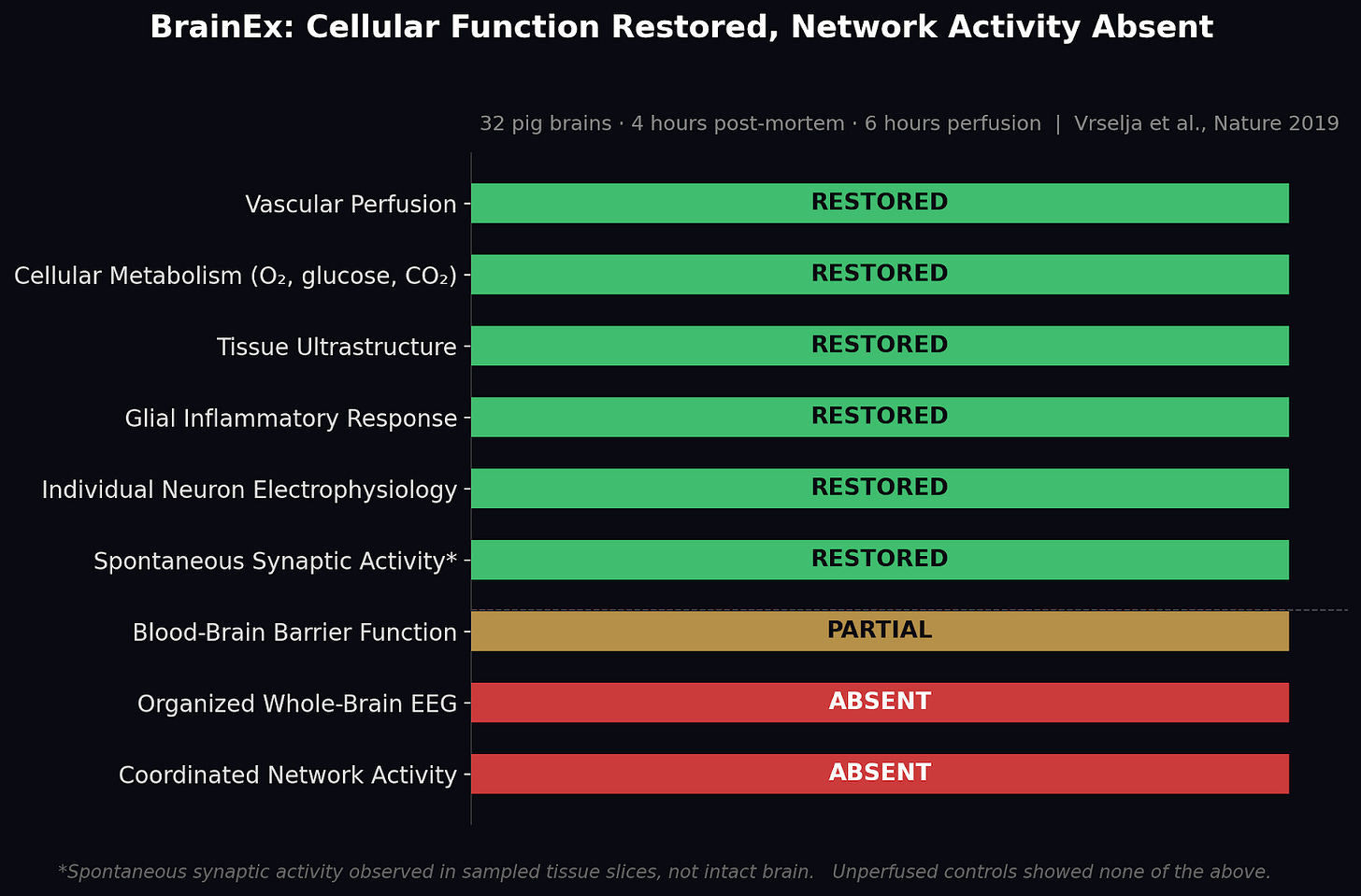
Vascular perfusion restored throughout the brain. Cells consumed oxygen, metabolized glucose, produced carbon dioxide. Tissue under electron microscopy was markedly intact compared to unperfused controls, which showed expected decomposition. Glial cells mounted inflammatory responses. The blood-brain barrier partially recovered. Individual neurons, when sampled and tested with electrodes, showed preserved electrophysiological properties and spontaneous synaptic activity.
But continuous EEG monitoring showed flatline throughout. Whatever was happening was cellular, not coordinated. Individual neurons worked. The networks connecting them did not.
Figure 2. BrainEx restored cellular-level function but no organized network activity was detected.
Then there are the neural activity blockers. The stated rationale is methodological: the experiment measured cellular viability, and spontaneous firing would consume energy and damage the tissue being studied. Fine. But the team also kept general anesthesia on standby and ran continuous EEG for the duration. You don’t monitor for something you’re certain won’t happen. The paper says nothing about what might occur without the blockers. Neither has anyone else. The absence of global activity wasn’t proof of irreversibility. It was a condition of the experiment. That line hasn’t been crossed.
Three years later, the same lab went whole-body. OrganEx perfused entire pigs one hour after cardiac arrest. Hearts regained electrical activity and contractile function. Livers produced albumin. Kidneys resumed filtration. Involuntary muscular contractions appeared in the head and neck, surprising even the researchers (Andrijevic et al., 2022). The cellular death cascade turned out to be slower, more complex, and more interruptible than anyone in clinical medicine had assumed.
Figure 3. From BrainEx (2019) to OrganEx (2022): expanding from brain to whole-body perfusion.
So what do you do with a technology that reverses death at the cellular level?
The near-term one is organ preservation. Thousands of donor organs are discarded annually because they can’t survive the gap between procurement and transplant. A system that restores cellular function after extended ischemia attacks that problem directly.
The longer-term one is drug discovery. Sestan and Vrselja co-founded Bexorg to do something no one else can: test drug candidates on perfused human brains — not organoids, not mouse models, but actual human tissue from donors with neurological disease — Alzheimer’s, Parkinson’s, the conditions where 90–99% of candidates fail. The academic science behind the platform is published in *Nature*. Bexorg has not published its own data. No FDA framework exists for this. The technology is real. The business case is unproven.
Neither application changes the core finding. Four hours of warm ischemia did not destroy these cells. The boundary between reversible and irreversible brain death is not where medicine placed it. That matters independent of whether anyone commercializes it.
**References**
Vrselja Z, Daniele SG, Silbereis J, et al. Restoration of brain circulation and cellular functions hours post-mortem. *Nature*. 2019;568(7752):336-343. PMID: 30918408
Andrijevic D, Vrselja Z, Lysyy T, et al. Cellular recovery after prolonged warm ischaemia of the whole body. *Nature*. 2022;608(7922):405-412. PMID: 35922506
Farahany NA, Greely HT, Giattino CM. Part-revived pig brains raise slew of ethical quandaries. *Nature*. 2019;568(7752):299-302.




This study is a stunning validation of Michael Levin’s framework of distributed agency. By using a liver to reboot a brain 50 minutes post-mortem, we see that the mind isn't just "in the head"—it is a property of the entire system's ability to maintain its informational state.
It suggests that our current definition of death is merely a technical limitation of our tools, not a biological finality. As Levin argues, as long as the multiscale competency of the cellular agents remains intact, death is simply a temporary decoherence of the collective intelligence. The liver didn't just filter blood; it provided the essential boundary conditions for that collective to re-emerge and remember how to function.
That thumbnail is sick!