BioWire Bytes 022 – Weight Loss Drugs May Prevent Neurodegenerative Disease. But They Don't Treat Them.
Paradoxical Data Shows GLP-1s Reduce Neurodegenerative Disorders Onset While Not Treating Them.
The fastest-growing drug class in the world was designed for the gut and obesity. The most important question about it may involve the brain and dementia.
First, if you enjoy these posts, consider subscribing and becoming a part of our growing community!
More than 50 million people worldwide now take GLP-1 receptor agonists for weight loss or diabetes. A growing number of researchers want to know what those weekly injections are doing to their brains. The largest systematic review and meta-analysis to date just tried to find out.
Choudhury and colleagues screened 10,037 records and included 82 studies covering Parkinson’s disease, Alzheimer’s disease, depression, anxiety, substance use disorders, binge eating, and suicidality (Choudhury, et al., 2026).
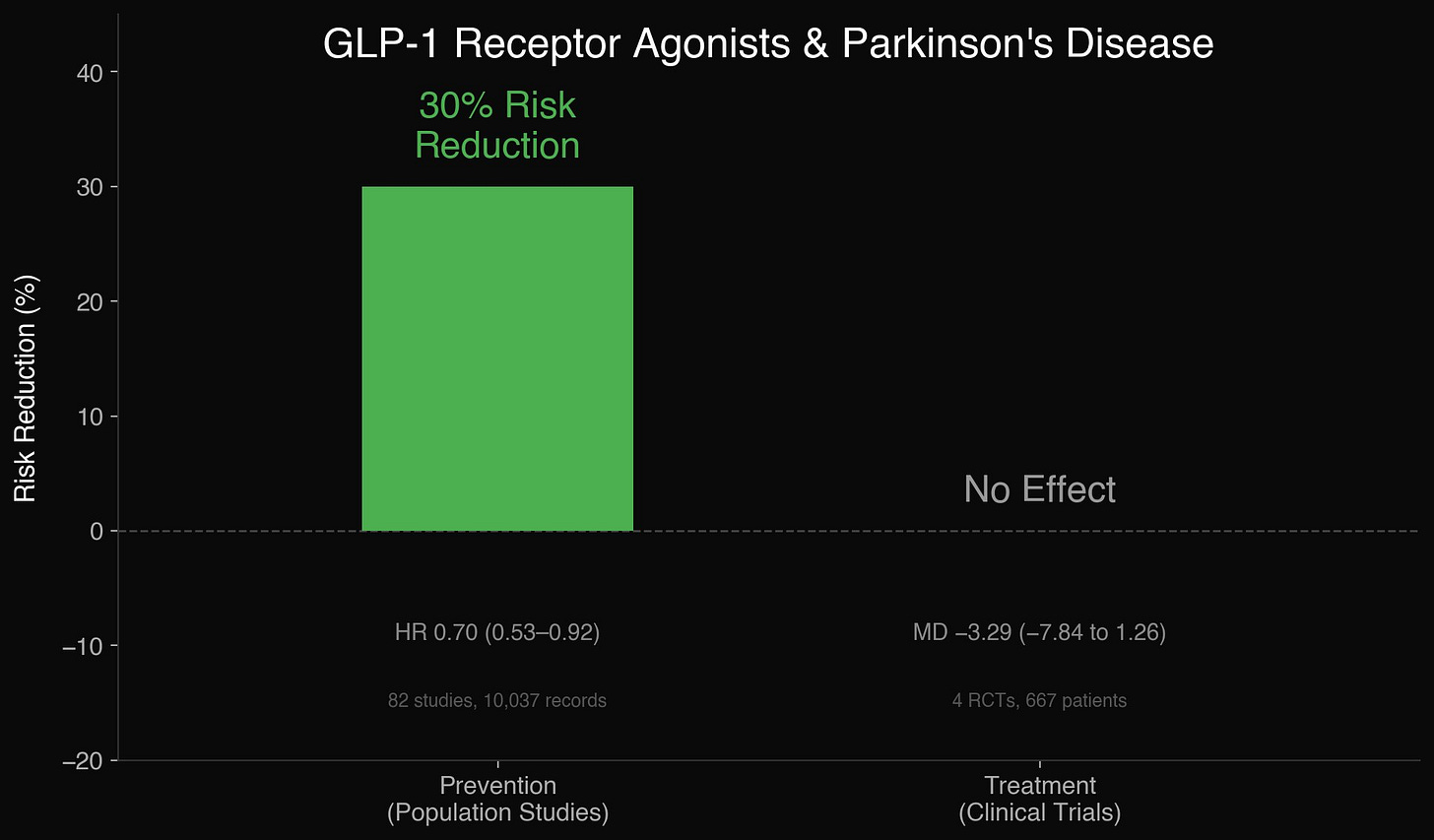
GLP-1 receptor agonist users had a 30% lower risk of developing Parkinson’s disease (HR 0.70). That’s a large signal from a drug class never designed to touch the basal ganglia. Study-to-study variability was moderate, which for observational data spanning multiple drug formulations is reassuring.
When Choudhury’s group looked at patients who already had Parkinson’s, the drugs didn’t help. No improvements in motor symptoms. No improvements in non-motor symptoms. No improvement in quality of life or dyskinesia scores. On the MDS-UPDRS Part III, the standard motor function scale, the drugs were indistinguishable from placebo.
A separate meta-analysis reinforces this. Stefanou and colleagues pooled all four randomized placebo-controlled trials of GLP-1 agonists in Parkinson’s, 667 patients across exenatide, lixisenatide, liraglutide, and NLY01, and found nothing (Stefanou, et al., 2026). No benefit on motor scores. No benefit on cognition. No benefit on non-motor symptoms. The drugs were well tolerated. They just didn’t work.
GLP-1 receptor agonists appear to prevent Parkinson’s disease but cannot treat it.
This prevention-without-treatment pattern exists elsewhere in medicine. Statins prevent heart attacks far more reliably than they reverse existing coronary artery disease. Aspirin prevents strokes more effectively than it rehabilitates stroke damage. The preventive signal is large enough to detect in population data. The treatment signal is zero.
Why? One explanation is direct neuroprotection. GLP-1 receptors are expressed throughout the brain, including in the substantia nigra, which degenerates in Parkinson’s. Some GLP-1 agonists cross the blood-brain barrier in animal models. Preclinical work shows they suppress neuroinflammation, reduce oxidative stress, and enhance mitochondrial function in dopaminergic neurons. Semaglutide in particular modulates microglial activation in rodent models of neurodegeneration (Evola and Parmar, 2026). The preclinical case is real. But it hasn’t translated to humans.
There’s a simpler explanation. GLP-1 agonists fix metabolism. They reduce blood sugar, visceral adiposity, insulin resistance, and systemic inflammation. Chronic systemic inflammation drives neuroinflammation through well-characterized pathways. Reduce the upstream metabolic dysfunction and you reduce the downstream inflammatory cascade that accelerates neuronal loss. Under this framing, GLP-1 agonists aren’t protecting the brain directly. They’re protecting the body, and the brain benefits.
The Alzheimer’s data is the strongest evidence for the metabolic explanation. GLP-1 RA users in Choudhury’s analysis showed a 63% reduced risk of Alzheimer’s compared to unmatched controls (RR 0.37). But when compared to SGLT2 inhibitors, another metabolically active drug class, the advantage vanished (RR 1.07). SGLT2 inhibitors don’t cross the blood-brain barrier. They don’t bind GLP-1 receptors. They fix metabolism through an entirely different mechanism. If both drug classes produce the same reduction in Alzheimer’s incidence, the simplest interpretation is that metabolic rescue is doing the work, not direct neuroprotection.
Novo Nordisk’s Phase 3 EVOKE and EVOKE+ trials reinforce this. The trials enrolled 3,808 participants with amyloid-confirmed early-stage Alzheimer’s and tested oral semaglutide against placebo for two years (Scheltens, et al., 2026). Semaglutide did not slow disease progression in either trial. Cerebrospinal fluid biomarkers of tau pathology and neuroinflammation improved modestly, around 10%, but none of that translated into cognitive or functional benefit. Novo Nordisk terminated the program. If semaglutide had meaningful direct neuroprotective effects in humans, you would expect some signal in 3,808 patients over two years. There was none on any clinical measure.
The pattern across both diseases is consistent. Observational data shows lower incidence of neurodegenerative disease among GLP-1 RA users. Randomized trials show no therapeutic effect once disease is established. The SGLT2 comparator suggests the prevention signal may not be specific to GLP-1 receptor activation at all. The most parsimonious explanation is that metabolic health protects the brain, and GLP-1 agonists happen to be the most widely prescribed way to achieve it.
Elsewhere, GLP-1 RAs were associated with a 45% reduced risk of cannabis use disorder and reduced binge eating frequency, but showed no effect on depression or anxiety. The suicidality question that has followed this drug class since FDA surveillance signals in 2023 found no increased risk in any analysis, with observational data trending protective.
Caveats apply. The prevention data is entirely observational, carrying the usual confounding and healthy-user biases. No randomized trial has tested whether GLP-1 agonists prevent neurodegenerative disease in healthy people. Evidence certainty ranged from moderate to very low depending on the outcome. Choudhury’s group calls the findings “hypothesis-generating.”
The real story here may not be that GLP-1 drugs protect the brain. It may be that metabolic health protects the brain. The drug class just happens to be the largest unintentional experiment in testing that hypothesis. Whether the prevention signal holds up in a prospective trial designed to isolate it remains an open question. No one has run that trial yet.
These newsletters take significant effort to put together and are totally for the reader’s benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Choudhury, I., Ward, J.H., Mahesh, S., Alam, U., Azmi, S. and Anson, M. (2026) ‘Effect of Glucagon-Like-Peptide-1 Receptor Agonists (GLP-1 RA) on Neuropsychiatric Outcomes: A Systematic Review and Meta-Analysis’, Clinical Therapeutics. doi: 10.1016/j.clinthera.2026.02.010.
Evola, V. and Parmar, M.S. (2026) ‘Targeting neuroinflammation in neurodegenerative disorders: the emerging potential of semaglutide’, Inflammation Research, 75(1), 13. doi: 10.1007/s00011-025-02166-6.
Scheltens, P., Atri, A., Feldman, H.H. et al. (2026) ‘Baseline characteristics from evoke and evoke+: Two phase 3 randomized placebo-controlled trials of semaglutide in participants with early-stage symptomatic Alzheimer’s disease’, Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 12(1), e70200. doi: 10.1002/trc2.70200.
Stefanou, M.I., Panagiotopoulos, E., Tentolouris, A. et al. (2026) ‘Efficacy and safety of glucagon-like peptide-1 receptor agonists in Parkinson’s disease: a systematic review and meta-analysis of randomized placebo-controlled clinical trials’, Therapeutic Advances in Neurological Disorders, 19, 17562864251408269. doi: 10.1177/17562864251408269.



Since 2020 I've been really suspicious of "X% less likely to" statements. Less likely than what? Is that the relative or absolute risk reduction? I majored in English, not statistics, and I'm not going to pretend that I can parse studies well enough to catch design errors or appreciate all the implications, but I know that when they don't report efficacy in ARR terms as well as RRR they're leaving out important context.