BioWire Bytes 014 - Weight-loss Drugs May Protect the Brain
Byte-sized Biotech
Another day, another study on secondary indications for “weight‑loss drugs.” At this point, that’s shorthand for GLP‑1 drugs like semaglutide and GLP‑1/GIP dual agonists like tirzepatide. What’s interesting is these drugs aren’t just helping diabetics with sugar control and celebrities lose weight; they may be quietly protecting the brain and vasculature. A new, large US EHR study in JAMA Network Open followed 60,860 matched adults with type 2 diabetes (with obesity) and found that starting semaglutide or tirzepatide was associated with lower risks of dementia, ischemic stroke, and all‑cause mortality versus other antidiabetic drugs. Let’s dive into the details.
First, if you enjoy these updates, consider subscribing and becoming a part of our growing community!
The study design.
Diabetes and obesity travel together more often than not—and both push dementia and stroke risk higher over time. The authors tapped the TriNetX US network, identified new users of semaglutide/tirzepatide, and matched them 1:1 to new users of other antidiabetic drugs on demographics, comorbidities, BMI, HbA1c, socioeconomic factors, and medication history. That left 30,430 patients per arm (mean age ~58; ~50% women) with a mean 1.8 years of follow‑up, and outcomes tracked out to seven years. They looked for first events: dementia, Parkinson’s, mild cognitive impairment, ischemic stroke, intracerebral hemorrhage, and also followed all‑cause mortality.
What turned up.
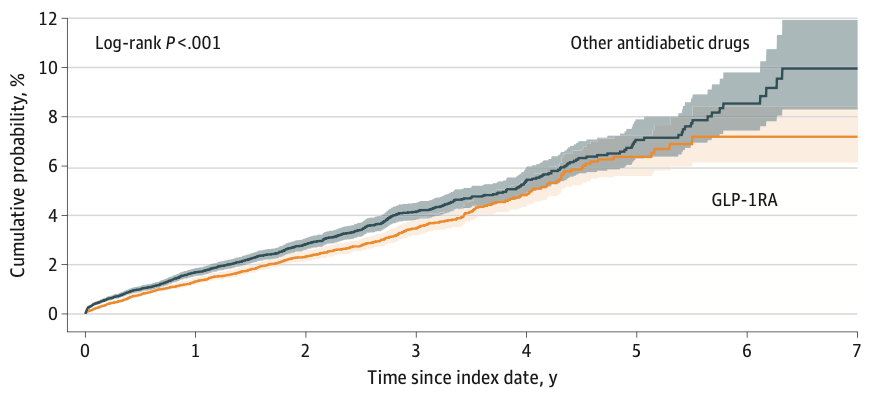
The curves didn’t sprint apart on day one. They drifted, then separated after ~1 year and kept widening. By year seven, the composite neuro/cerebrovascular outcome was 7.2% for GLP‑1 users vs 10% for peers on other drugs. In plain English: fewer bad brain/vascular outcomes over time for the GLP‑1 group.
Some punchlines:
Dementia: GLP-1RA users had 37% lower risk. The subtype signal was strongest for “other dementia”; AD/vascular dementia estimates were not significant, likely power-limited.
Stroke: 19% lower risk of ischemic stroke.
Mortality: 30% lower all-cause mortality.
Subgroups: Signals were more pronounced in ≥60 years, women, and BMI 30–40.
Agent-specific hints: In stratified analyses, semaglutide carried the dementia reduction (HR 0.63 vs comparators), while tirzepatide showed a sharper stroke reduction (HR 0.69) and a notable mortality signal (HR 0.48). Treat as exploratory.
No effect on PD: Parkinson’s disease risk was unchanged; mild cognitive impairment was also not reduced.
Why this is believable.
The cardiometabolic wins of GLP‑1s have been well documented, and stroke hints popped up before in other clinical studies (SUSTAIN‑6 / PIONEER‑6). On the biology side, GLP‑1 signaling has been shown to cool neuroinflammation, improve endothelial function, and touch amyloid/tau pathways in models, exactly the axes you’d want to move for dementia and ischemic disease. That coherence helps explain why we see ischemic stroke and dementia move, but not hemorrhagic stroke. The paper also notes that clinical studies EVOKE / EVOKE+ are in progress to test semaglutide directly in early Alzheimer’s, which will be decisive.
The fine print.
This is an observational study from electronic health record (EHR) data. It has a good design (new‑user, active comparator, extensive matching), but there’s lots of potential confounders (frailty, adherence, dosing). The dosages were inferred from prescriptions, biomarker/imaging data aren’t available, and TriNetX doesn’t support classic competing‑risk methods with death (the authors partially addressed that with composites and a separate mortality analysis). This is strong evidence, but it’s not the same definitive proof you get from a double-blinded placebo-controlled clinical study.
What to watch next.
The Phase 3 (EVOKE/EVOKE+) readouts in Alzheimer’s, and other trials in high‑risk metabolic populations that track dose/adherence, add fluid biomarkers and neuroimaging, and apply full competing‑risk methods. That is the evidence that will turn this from “promising association” to “practice guidance”.
These newsletters take significant effort to put together and are totally for the reader's benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Lin HT, Tsai YF, Liao PL, Wei JCC. Neurodegeneration and Stroke After Semaglutide and Tirzepatide in Patients With Diabetes and Obesity. JAMA Network Open. 2025;8(7):e2521016. July 15, 2025. Open access.


I wonder if all of these benefits are related to metabolism / caloric restriction.
Mind blown 🤯