BioWire Bytes 007 - New Hair Growth Drug PP405 Shows Promise in Clinic
Byte-sized biotech
The appearance and health of our hair plays a deep role in our individual identity, making its loss deeply traumatic. Unsurprisingly, people have been searching for a treatment for hair loss for centuries. The wackiest historical remedy I've come across was an ancient Egyptian concoction combining hippopotamus fat, crocodile dung, and ibis droppings. Maybe this was reserved for a less-than-popular Pharaoh! Yet, even in today’s world of advanced therapeutics and artificial intelligence, our treatments remain surprisingly limited. Go-to drugs like minoxidil (Rogaine) and finasteride (Propecia) only help, at best, one in three hair-loss sufferers, mostly slowing down hair loss or maintaining existing strands rather than regrowing brand-new hair. It's been, frankly, pretty underwhelming.
Now, a new approach emerging from UCLA appears to be much more exciting, not just preserving existing hair, but potentially reversing hair loss altogether. It works by flipping a metabolic "on" switch in dormant follicles. The experimental topical drug, dubbed PP405 (a cheeky nod to Los Angeles’s famously congested 405 freeway), is designed to reawaken dormant hair follicle stem cells, stimulating them to grow hair again. Early results have scientists cautiously optimistic that we might finally have a genuine hair-regrowth therapy on the horizon.
A Metabolic Strategy to Restart Hair Follicles
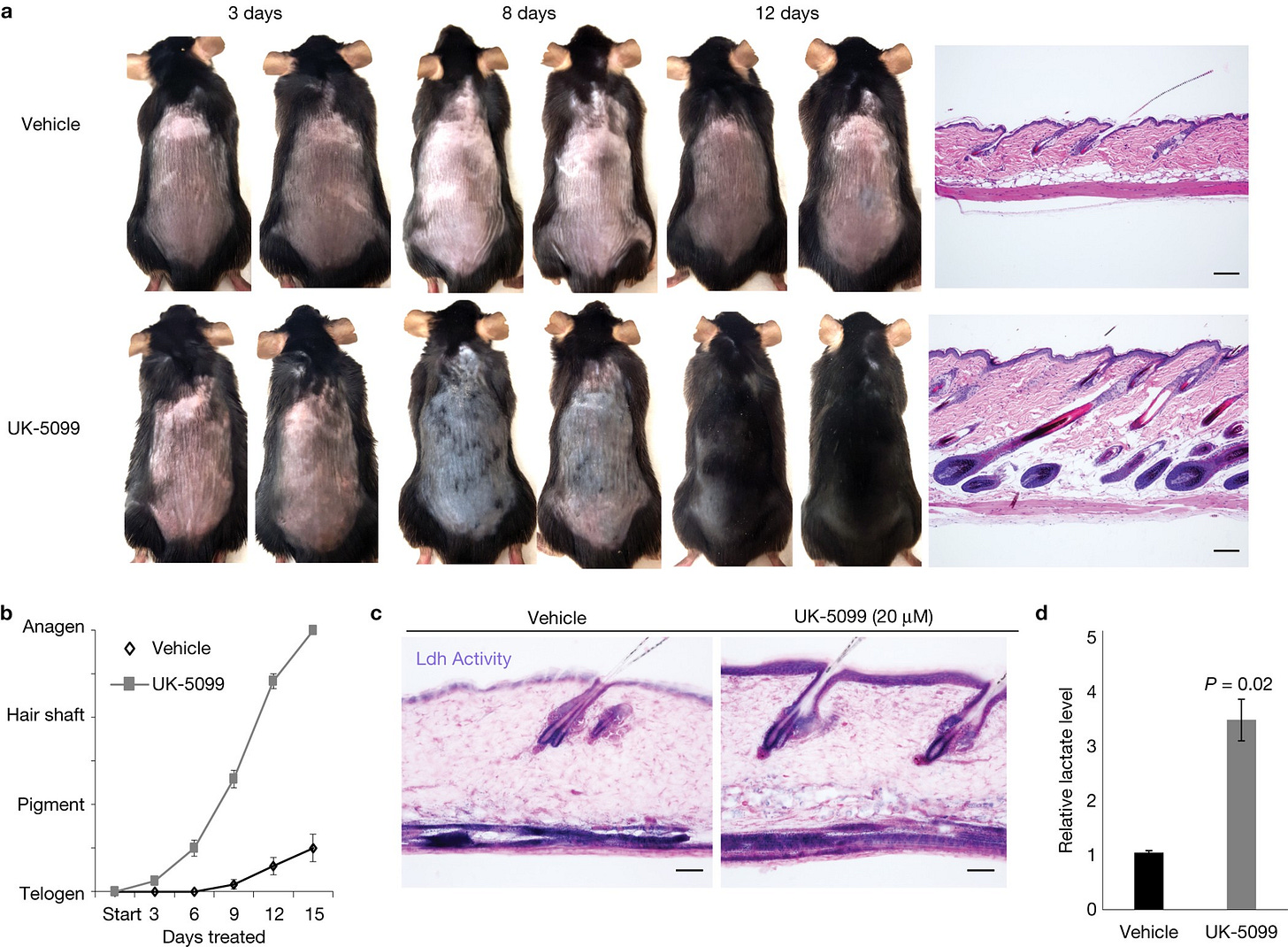
PP405’s secret weapon is metabolism. It works by inhibiting the mitochondrial pyruvate carrier (MPC), essentially blocking a key fuel pipeline into the cell’s energy factories (mitochondria). When denied their usual fuel route, the follicle’s stem cells switch to an alternative energy mode and ramp up glycolysis, a faster but less efficient way of making energy (the same process that makes your leg muscles “feel the burn” during an intense sprint). This metabolic pivot isn’t about producing more energy per se, but about creating the right biochemical signals to jump-start growth. By forcing cells to churn through sugar outside mitochondria, PP405 causes a buildup of lactate and alters the NAD⁺/NADH ratio, a shifts that act like a molecular wake-up call for dormant stem cells. Once metabolically reactivated, these stem cells can exit their dormant state and start generating new hair shafts. It’s a fundamentally different strategy than anything we’ve had before, essentially rebooting the follicle’s growth engine rather than externally prodding it with hormones or blood-flow enhancers.
Not Your Average Hair Loss Remedy
PP405 is delivered in a similar way, as a once-daily topical solution, but it works via metabolism rather than hormones or circulation.
Unlike Rogaine or Propecia, PP405 doesn’t rely on hormones or blood vessels, and that’s a big deal. Minoxidil (Rogaine) stimulates hair partly by boosting scalp blood flow and prolonging the growth phase of hairs, while finasteride (Propecia) works hormonally by blocking DHT (a testosterone-derived hormone that shrinks follicles). Those methods can help some people, but they come with limitations: finasteride’s hormone interference can cause very unwelcome side effects, like libido changes, and minoxidil often yields only fine “peach fuzz” regrowth in many users. PP405’s non-hormonal method directly acts on the target cells with a local application, and because it’s topically applied to the scalp as a gel or solution and engineered to stay local, PP405 also avoids significant absorption into the bloodstream. That means a lower risk of body-wide side effects. Another perk is that, since PP405 works via a unique metabolic pathway, meaning it could potentially be combined with other existing treatments (imagine pairing it with minoxidil, low-level laser therapy, or even finasteride) to attack hair loss from multiple angles. In short, PP405 is a new class of hair loss therapy focused on follicle stem cell revival.
Early Results: From Mice to Men
How did all of this start? The first hints of PP405’s promise came from laboratory mice. Researchers at UCLA found that blocking the MPC “fuel pipeline” in mouse follicles was like hitting a growth switch; previously dormant follicles started cycling again, producing healthy new hairs within a month. This 2017 mouse study, published in Nature Cell Biology, laid the scientific groundwork by showing that tweaking metabolism alone could spur hair regeneration (Flores et al., 2017). Building on that success, the UCLA team moved PP405 into human testing. In a small Phase 1 trial in 2023, 20 men with thinning hair applied PP405 to their scalps each night for just one week. Even in that short span, the drug showed signs of waking up follicles. Men treated with PP405 started sprouting new terminal hairs (the thick, pigmented kind) rather than just barely-visible fuzz, a key proof of concept. Importantly, there were no serious side effects; the compound stayed localized in the scalp and did not measurably enter the bloodstream, and participants reported no hormonal disturbances. Dr. Michael Jung, the chemist who helped design PP405, intentionally gave the molecule properties to keep it trapped in the skin so it can do its work on follicles without roaming the body.
Building on the Phase 1 safety success, Pelage Pharmaceuticals (the startup furthering this research) launched a larger Phase 2a trial in 2024 to get early efficacy data. In this placebo-controlled study, ~78 men and women applied either PP405 or a placebo daily for 4 weeks, with follow-up out to 12 weeks. The headline result? Even with just one month of dosing, PP405 showed a convincing hair boost in a subset of patients. By 8 weeks (about a month after stopping treatment), 31% of treated men with significant hair loss saw at least a 20% increase in hair density, whereas 0% of the placebo group hit that threshold. In other words, roughly one-third of the men grew noticeably more hair in just weeks. For reference, visible results often take 6–12 months in other treatment modalities. Even more exciting, doctors noted that some new hairs emerged in previously bald areas, not just where existing thin hairs were hanging on. This is more evidence that PP405 is truly resurrecting dormant follicles that had long ago given up, rather than merely thickening existing hair. It’s still early data from a relatively small trial, but these findings are exciting and provide early validation of PP405’s regenerative potential. Equally crucial, the Phase 2a study reaffirmed PP405’s safety: no systemic absorption was detected and no major side effects were reported, meaning the topical is behaving as intended. All together, the preclinical and clinical results so far are strong indicators that PP405 is a fast-acting, well-tolerated agent that actually generates new hair, something current treatments struggle to achieve.
How did this work go from an academic lab to a vc-backed startup?
It’s worth noting that PP405’s journey from campus lab to clinical trials has been remarkably swift. The molecule was born from a decade of stem cell research at UCLA, led by scientists Dr. William Lowry, Dr. Heather Christofk, and chemist Dr. Michael Jung. After their 2017 breakthrough showed metabolism could trigger hair growth, these researchers teamed up to found a startup, Pelage Pharmaceuticals, in 2018. (The name “Pelage” fittingly means animal fur or coat – a nod to hair.) UCLA granted Pelage an exclusive license to develop the MPC-inhibiting compounds for hair loss, and the company quickly attracted investors. In 2023, Google Ventures (GV) led a $16–17 million Series A financing to bankroll PP405’s initial clinical trials (maybe there are a lot of software engineers looking for hair remedies?) Other biotech VCs joined in as well, betting that Pelage’s fresh approach could address an enormous market of balding consumers. The partnership between academia and venture capital has provided Pelage the resources to run trials at a good clip – Phase 1 and 2 were completed back-to-back within about two years. Notably, even pharmaceutical giant Allergan took interest early on, signing an option in 2019 to acquire Pelage down the line (though that option was later dropped after corporate changes). Today, Pelage remains independent and focused on pushing PP405 through the final phases of testing. The company’s leadership includes dermatology experts from Harvard/Mass General and seasoned biotech executives, underscoring that this is a serious scientific effort, not just a cosmetic fad. With big-name backers and a clear biological rationale, PP405 has quickly moved from a university discovery to one of the most closely watched new therapies in the hair loss field.
What comes next?
Despite the buzz, PP405 is still in its experimental phase, and key questions remain regarding safety and efficacy. The Phase 2a trial was relatively short, we don’t yet know how much regrowth is truly achievable with longer treatment, or how sustained the benefits will be. Will those revived follicles keep producing hair long-term, or will they fall dormant again without continuous use? Moreover, can you deplete these stem cells? It’s also unclear how well PP405 will work for people with very advanced baldness (who may have fewer dormant follicles left to rescue) or for those who’ve been bald for many years. The early trials skewed toward men; ongoing studies are including women, but results in female pattern hair loss still need more data. Safety over the long haul is another consideration. So far, PP405’s local delivery looks clean, but only larger, longer trials can definitively rule out rare side effects. All these unknowns mean that cautious optimism is the right attitude for now. As Dr. Lowry of UCLA frankly noted, “No such product will work for everyone,” even if this metabolic approach is a major leap forward.
The good news is that we won’t have to wait too long to get more answers. Pelage plans to kick off Phase 3 trials in 2026, enrolling a much larger population of men and women to more rigorously test PP405’s efficacy and safety. Phase 3 will tell us just how well PP405 performs against a placebo over a longer period and whether those impressive early hair gains hold up. If all goes well, regulatory approval could follow a couple of years thereafter. The company’s roadmap suggests a hopeful timeline: full Phase 2 results by 2025–26, Phase 3 completion by ~2027, and – in a best-case scenario – an FDA greenlight for PP405 by the late 2020s. That would make it the first truly novel hair loss drug in a generation. Of course, drug development can be unpredictable, and anything can happen in Phase 3. But the mere fact that a metabolic stimulator like PP405 has come this far is itself a sign of progress in this field. After decades of stagnation in hair loss remedies, we’re finally seeing a therapy that generates new hair rather than just hanging onto old hair.
Would you try this product? Comment your answer below!
These newsletters take significant effort to put together and are totally for the reader's benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
https://clinicaltrials.gov/study/NCT06393452?term=pp405&rank=1
https://pelagepharma.com/
https://www.businesswire.com/news/home/20250617338859/en/Pelage-Pharmaceuticals-Announces-Positive-Phase-2a-Clinical-Trial-Results-for-PP405-in-Regenerative-Hair-Loss-Therapy
https://www.dermatologytimes.com/view/q-a-pelage-s-novel-pp405-advances-to-phase-2a-for-androgenetic-alopecia
Flores, A., Schell, J., Krall, A.S., Jelinek, D., Miranda, M., Grigorian, M., Braas, D., White, A.C., Zhou, J.L., Graham, N.A. and Graeber, T., 2017. Lactate dehydrogenase activity drives hair follicle stem cell activation. Nature cell biology, 19(9), pp.1017-1026.



i wanna try it on a woolly mammoth 🤭
As someone who would benefit from this, this is very interesting. Thanks for all the details on the mechanism and biochemistry, David.