A Single Injection Replaced the Entire CAR-T Pipeline
A single intravenous dose generated cancer-killing immune cells without cell harvesting, manufacturing, or chemotherapy.
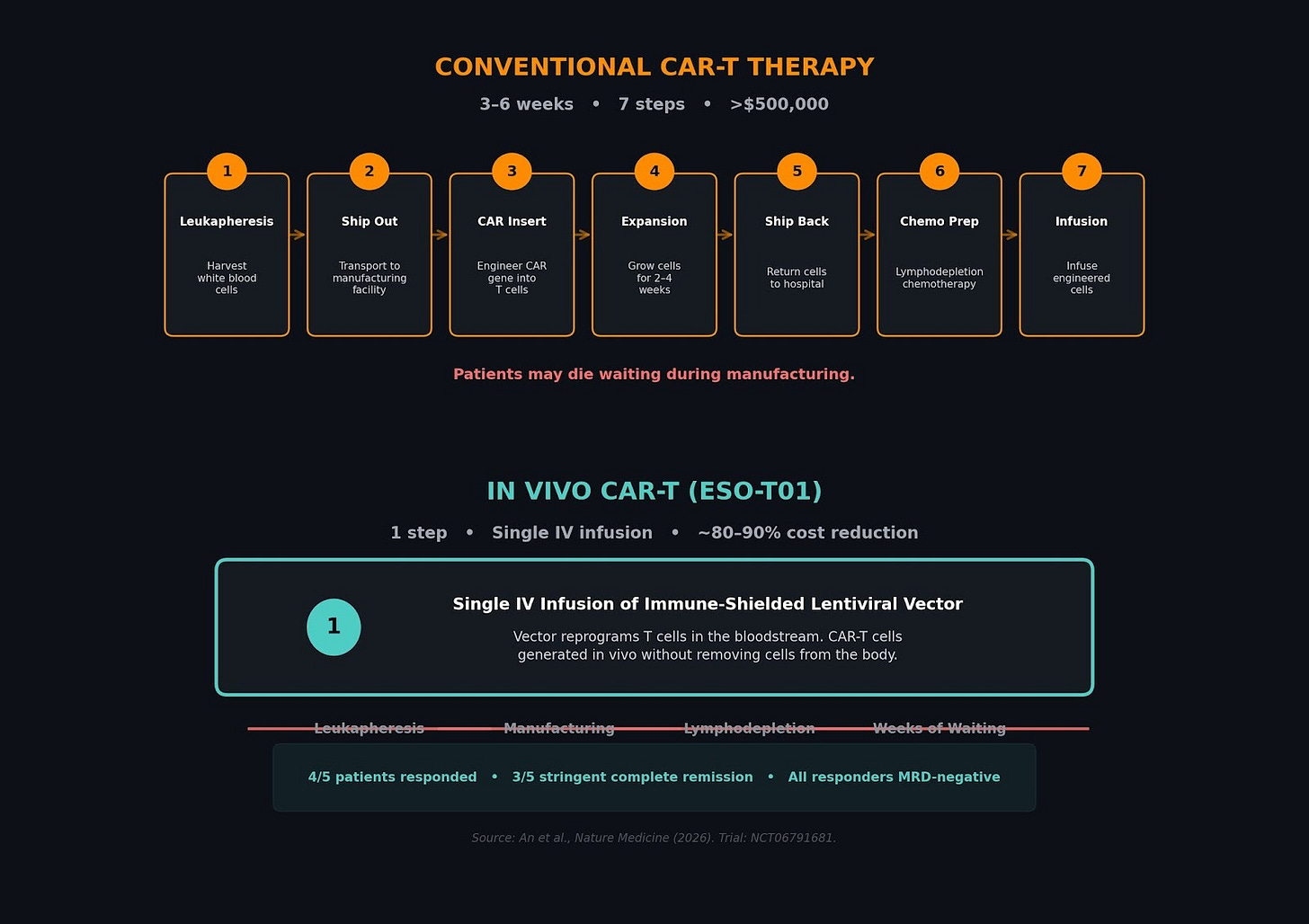
Blood cancers were once a death sentence. Two CAR-T therapies approved since 2021 pushed overall response rates past 70 percent in relapsed multiple myeloma (Munshi et al., NEJM, 2021; Berdeja et al., Lancet, 2021). The problem is everything that comes before the infusion. These treatments take three to six weeks to build, cost half a million dollars, and require lymphodepleting chemotherapy before they even begin.
For these treatments, doctors harvest a patient’s white blood cells through leukapheresis. Those cells ship to a manufacturing facility where cell engineers reprogram them with a chimeric antigen receptor, a synthetic receptor that recognizes a specific cancer protein. The reprogrammed cells are expanded in culture for weeks. Meanwhile, the patient undergoes lymphodepleting chemotherapy to clear space in the immune system. Then the engineered cells are infused back into the patient. The total bill exceeds $500,000 per patient (American Cancer Society, 2025; Rawlings et al., Clin Hematol Int, 2024). Patients routinely deteriorate or die during the wait.
If you enjoy these posts, consider subscribing and becoming a part of our growing community!
A phase 1 trial out of Tongji Hospital in Wuhan just cut all of that out and skipped to the life-saving part (An et al., Nature Medicine, 2026).
The treatment is ESO-T01, an immune-shielded lentiviral vector that carries the genetic instructions for an anti-BCMA chimeric antigen receptor. BCMA is a protein that sits on the surface of myeloma cells. The vector reprograms T cells directly in the bloodstream, turning the patient’s own immune cells into cancer killers without ever removing them from the body.
Five patients with relapsed or refractory multiple myeloma received a single intravenous infusion. No leukapheresis for immune cell isolation. No ex vivo cell engineering or multiweek expansion. No lymphodepleting chemotherapy. Just one injection.
Figure 1. Conventional CAR-T therapy requires seven steps over 3–6 weeks at a total cost exceeding $500,000. The in vivo approach (ESO-T01) reduces this to a single intravenous infusion. Source: An et al., Nature Medicine (2026).
The goal of a phase 1 study is to collect safety data. The efficacy results were not supposed to be the story.
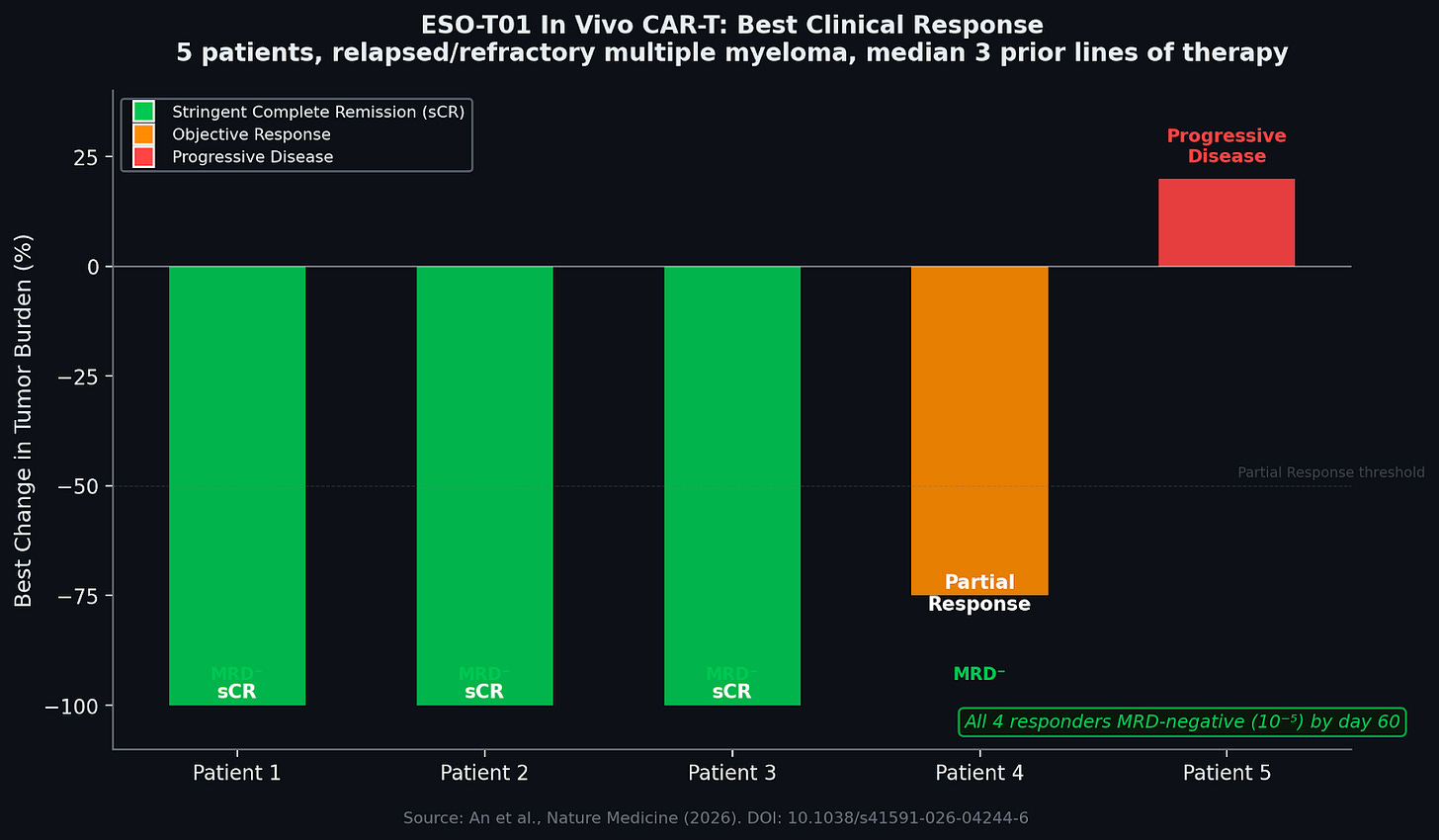
Four of five patients achieved objective responses. Three achieved stringent complete remission, the deepest category of response in myeloma. The approved therapies that achieve those response rates require leukapheresis, weeks of manufacturing, and lymphodepleting chemotherapy. This study matched them with a single injection and none of it.
All four evaluable responders were minimal residual disease negative at the 10-5 sensitivity level by day 60. That means fewer than one myeloma cell per 100,000 bone marrow cells. Those numbers belong in a phase 3 readout. This is phase 1.
Figure 2. Clinical responses in the ESO-T01 phase 1 trial. Three of five patients achieved stringent complete remission (sCR). All four responders were MRD-negative at 10-5 sensitivity by day 60. Source: An et al., Nature Medicine (2026).
The economics of CAR-T therapy are built on infrastructure. Manufacturing alone represents roughly 75 percent of total treatment cost (Hernandez et al., JAMA Oncol, 2018).
Building the weapon inside the body eliminates most of that. No manufacturing facility. No weeks of inpatient stays. The drug itself, a viral vector produced at scale, behaves more like a conventional biologic than a bespoke cell therapy.
No one has published a formal cost estimate for in vivo CAR-T yet. A KPMG analysis modeled how manufacturing simplification could bring the cost below $30,000 per treatment (KPMG, 2024). In vivo generation is the most radical version of that simplification.
If you can build it inside, the infrastructure collapses. CAR-T therapy currently exists at roughly 200 centers worldwide, nearly all in the United States, Europe, and a handful of Chinese cities. The majority of the world’s cancer patients have zero access. A viral vector that ships at scale and requires no cell manufacturing facility changes that. A patient in rural Arkansas, or Lagos, or rural India, could receive a single IV infusion at a community clinic.
A cancer treatment that once required a factory now requires a syringe.
The field has not fully reckoned with another advantage: repeat dosing. Ex vivo CAR-T is functionally a one-shot therapy. If the cancer relapses, re-manufacturing is difficult or impossible because the patient’s T cells are already depleted from the first round. An in vivo platform changes that calculus. If the first dose fails or the disease returns, the patient gets another injection. That opens the door to combination protocols: sequential targets, dose escalation, CAR-T paired with checkpoint inhibitors. Strategies that are financially prohibitive when each dose costs half a million dollars.
The safety data tell a more complicated story. Cytokine release syndrome, a systemic inflammatory response triggered when engineered immune cells activate and attack cancer en masse, occurred in four patients, three of them grade 3. All were controlled with standard interventions: corticosteroids, tocilizumab, supportive care (An et al., 2026). The most common side effects were transient drops in blood counts and reversible liver enzyme elevations. One patient died from spinal cord compression caused by an extramedullary myeloma lesion, a known complication of the underlying disease.
The trial enrolled five patients, all male, all heavily pretreated with a median of three prior lines of therapy. Median follow-up was six months. The trial was stopped early in 2025 for reasons the authors do not disclose. Phase 1 trials are designed to establish safety, not efficacy. The sample is too small for anything definitive, and durability of response remains an open question.
In 2003, a gene therapy trial for X-linked severe combined immunodeficiency used gamma-retroviral vectors to insert a corrective gene into patient cells. Several children developed leukemia. The vectors had integrated near proto-oncogenes and activated them (Hacein-Bey-Abina et al., Science, 2003). That episode set the entire field back by a decade.
Lentiviral vectors were engineered specifically to avoid this. Modern self-inactivating designs delete the enhancer and promoter elements that caused the problem, sharply reducing the risk of activating nearby genes after integration (Naldini, Nature, 2015). The clinical track record now supports the redesign. Over 500 patients have received lentiviral gene therapy across a dozen disease indications. The longest follow-up data, now past a decade, show no vector-related cancers (Scala et al., Nature Medicine, 2023; Kohn et al., NEJM, 2025). No confirmed case of replication-competent lentivirus has been reported in any patient to date (Merten et al., Mol Ther Methods Clin Dev, 2024).
But ESO-T01 is doing something those therapies did not. It is injecting the vector directly into the bloodstream of a patient who has not been lymphodepleted. Previous attempts at in vivo CAR-T generation struggled precisely here. The patient’s immune system neutralized the viral vector before it could reprogram enough T cells. ESO-T01 solves this with a nanobody-directed, immune-shielded envelope that evades complement and antibody recognition (An et al., 2026). That shielding is the key engineering innovation. Without it, the concept does not work.
The entire logistical architecture of CAR-T therapy exists because of one assumption: you have to build the weapon outside the body. Seven FDA-approved CAR-T products. Billions of dollars in manufacturing infrastructure. All built on that premise.
Other groups have pursued in vivo CAR-T using lipid nanoparticles or engineered fusogens, but ESO-T01 is the first to report clinical data in humans.
The platform is not specific to myeloma. The immune-shielded lentiviral vector is a chassis. Swap the receptor target and the same architecture could reprogram T cells to hunt different cancers, from B-cell lymphomas to solid tumors that have resisted every prior generation of CAR-T.
But cancer might not even be the most interesting application. In 2022, a team at the University of Erlangen gave ex vivo CAR-T cells to five patients with refractory systemic lupus erythematosus. All five went into drug-free remission (Mackensen et al., Nature Medicine, 2022). The engineered cells depleted the autoreactive B cells driving the disease. Similar results have since emerged in multiple sclerosis and systemic sclerosis. The limiting factor is the same one that limits cancer patients: manufacturing complexity, cost, and access.
An in vivo platform eliminates all three. A rheumatologist in a community clinic could administer a single infusion that reprograms the patient’s own T cells to clear the B cells attacking their joints, kidneys, or nervous system. No referral to an academic center. No six-figure bill. No three-week manufacturing wait while the disease progresses.
Five patients is a proof of concept, not a product. Larger trials will determine whether the efficacy holds and whether the safety profile is tolerable at scale.
But the question the field has been asking for a decade, whether you can skip the factory and program the immune system in place, now has preliminary human data behind it. The answer appears to be yes.
These newsletters take significant effort to put together and are totally for the reader’s benefit. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
An N, Wang D, Zhang P, et al. In vivo generation of anti-BCMA CAR-T cells in relapsed or refractory multiple myeloma: a phase 1 study. Nature Medicine (2026). DOI: 10.1038/s41591-026-04244-6.
Munshi NC, et al. Idecabtagene vicleucel in relapsed and refractory multiple myeloma. N Engl J Med 384, 705-716 (2021).
Berdeja JG, et al. Ciltacabtagene autoleucel in patients with relapsed or refractory multiple myeloma (CARTITUDE-1). Lancet 398, 314-324 (2021).
Hacein-Bey-Abina S, et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 302, 415-419 (2003).
Naldini L. Gene therapy returns to centre stage. Nature 526, 351-360 (2015).
Scala S, et al. Dynamics of genetically engineered hematopoietic stem and progenitor cells after autologous transplantation in humans. Nature Medicine 29, 1769-1780 (2023).
Kohn DB, et al. Lentiviral gene therapy with autologous hematopoietic stem and progenitor cells for adenosine deaminase-deficient SCID. N Engl J Med (2025).
Merten OW, et al. Long-term stability of clinical-grade lentiviral vectors. Mol Ther Methods Clin Dev 32, 101176 (2024).
American Cancer Society. CAR T-cell Therapy and Its Side Effects. cancer.org (2025).
KPMG. The USD 30k CAR-T Therapy: A Future Within Reach? (2024).
Hernandez I, et al. Total costs of chimeric antigen receptor T-cell immunotherapy. JAMA Oncol 4, 994-997 (2018).
Rawlings MO, et al. Navigating the economic burden of multiple myeloma. Clin Hematol Int (2024).
13. Mackensen A, et al. Anti-CD19 CAR T cells for refractory systemic lupus erythematosus. Nature Medicine 28, 2124-2132 (2022).
ClinicalTrials.gov: NCT06791681.


I'd really like to know why they stopped the trial early, and the fact that they didn't disclose that makes me want to know even more.
This would be transformational in our field. This would, in theory, make CAR-T available to community sites.